


Contents

This edition first published 2011, © 1980, 1984, 1993, 2001, 2006, 2011 by AV Hoff brand and PAH Moss
Blackwell Publishing was acquired by John Wiley & Sons in February 2007. Blackwell’s publishing program has been merged with Wiley’s global Scientific, Technical and Medical business to form Wiley-Blackwell.
Registered office: John Wiley & Sons Ltd, The Atrium, Southern Gate, Chichester, West Sussex, PO19 8SQ, UK
Editorial offices: 9600 Garsington Road, Oxford, OX4 2DQ, UK
The Atrium, Southern Gate, Chichester, West Sussex, PO19 8SQ, UK
111 River Street, Hoboken, NJ 07030-5774, USA
For details of our global editorial offices, for customer services and for information about how to apply for permission to reuse the copyright material in this book please see our website at
The right of the author to be identified as the author of this work has been asserted in accordance with the UK Copyright, Designs and Patents Act 1988.
First published 1980
Reprinted 1981, 1982, 1983 (twice)
Second edition 1984
Reprinted 1985
Reprinted with corrections 1985, 1988 (twice), 1989
German edition 1986 (reprinted 1996)
Japanese edition 1986
Spanish edition 1987 (reprinted twice)
Indonesian edition 1987
Third edition 1993
German 1996
Hungarian edition 1997
Chinese edition 1998
Reprinted with corrections 1993, 1994, 1995, 1996, 1997, 1998, 1999, 2000
Fourth edition 2001
German 2002
Indonesian 2005
Korean 2005
Portuguese 2005
Fifth edition 2006
Reprinted with Gaucher’s disease 2008
All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted, in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, except as permitted by the UK Copyright, Designs and Patents Act 1988, without the prior permission of the publisher.
Designations used by companies to distinguish their products are often claimed as trademarks. All brand names and product names used in this book are trade names, service marks, trademarks or registered trademarks of their respective owners. The publisher is not associated with any product or vendor mentioned in this book. This publication is designed to provide accurate and authoritative information in regard to the subject matter covered. It is sold on the understanding that the publisher is not engaged in rendering professional services. If professional advice or other expert assistance is required, the services of a competent professional should be sought.
The contents of this work are intended to further general scientific research, understanding, and discussion only and are not intended and should not be relied upon as recommending or promoting a specific method, diagnosis, or treatment by physicians for any particular patient. The publisher and the author make no representations or warranties with respect to the accuracy or completeness of the contents of this work and specifically disclaim all warranties, including without limitation any implied warranties of fitness for a particular purpose. In view of ongoing research, equipment modifications, changes in governmental regulations, and the constant flow of information relating to the use of medicines, equipment, and devices, the reader is urged to review and evaluate the information provided in the package insert or instructions for each medicine, equipment, or device for, among other things, any changes in the instructions or indication of usage and for added warnings and precautions. Readers should consult with a specialist where appropriate. The fact that an organization or Website is referred to in this work as a citation and/or a potential source of further information does not mean that the author or the publisher endorses the information the organization or Website may provide or recommendations it may make. Further, readers should be aware that Internet Websites listed in this work may have changed or disappeared between when this work was written and when it is read. No warranty may be created or extended by any promotional statements for this work. Neither the publisher nor the author shall be liable for any damages arising herefrom.
Library of Congress Cataloging-in-Publication Data
Hoffbrand, A. V.
Essential haematology / A.V. Hoff brand, P.A.H. Moss, – 6th ed.
p.; cm.
Includes bibliographical references and index.
ISBN 978-1-4051-9890-5
1. Blood–Diseases. 2. Hematology. I. Moss, P. A. H. II. Title.
[DNLM: 1. Hematologic Diseases. WH 120 H698e 2011]
RC633.H627 2011
616.1’5–dc22
2010024521
Preface to the Sixth Edition
Haematology has advanced more rapidly in the last ten years more than any branch of medicine. Current haematological literature is so prolific that it is increasing difficult for any one but a specialist to keep up to date.
The Anaemias by Janet Vaughan, 1st edition, Oxford Medical Publications, 1933
Almost 70 years later, haematology still continues to be at the forefront of medical advances. The increased understanding of blood diseases particularly their genetic basis and changes in their treatment is such that in writing this new edition, substantial changes have been necessary throughout. The classification of the neoplasms of the haemopoietic and lymphoid diseases has been revised by WHO (2008) and the names and definitions of many of these diseases have changed. Clinical features, genetics and immunophenotype are increasingly used to define biological entities. We have made changes in all the relevant chapters but, in a book intended primarily for undergraduates, we have simplified some of the classification tables and omitted detailed descriptions of rare diseases. On the other hand, some tests e.g. red cell survival and vitamin B12 absorption studies have become obsolete and are now omitted. As previously, we have used a colour line in the margin to indicate text that we consider more advanced than is needed for under graduate medical students and more appropriate for postgraduates.
John Pettit, co-author on all five previous editions, has retired from authorship for this edition. Much of the success of the book when it first appeared 30 years ago and in all five previous editions has been due to John’s ability to write clear, concise descriptions of the various diseases, and to produce first-class photomicrographs and line diagrams to illustrate the text. Many of these images appear in this latest edition.
The different aspects of iron overload are now merged into a new chapter and we have separated chapters on acute myeloid and acute lymphoblastic leukaemia. We have also introduced summary boxes at the end of each chapter to summarise the contents and added multiple choice questions to the website both at undergraduate and at a more advanced level to help in self-learning. The book’s website will be updated annually.
We would like to thank Elsevier for the use of the following figures: 4.2, 11.14, 13.5b, 18.5, 18.6, 18.7, 20.13–16, 20.18, 21.2b, 22.3, 23.13, 26.7, 30.5 from Hoffbrand A.V., Pettit J.E. and Vyas P. (2010) Color Atlas of Clinical Hematology, 4th edition. Mosby Elsevier, Philadelphia. We would also like to thank Professor John W. Weisel for the use of the chapter title figure from Brown A.E.X., Nagaswami C., Litvionov R.I. and Weisel J.W. (2009) Focusing on fibrin. Science 327: 741. The image shows colourised scanning electron micrograph of a thrombus taken from a patient with acute myocardial infarction. The thrombus is made up of a fibrin meshwork (brown) together with platelets (light purple). Erythrocytes (red) and leucocytes (green) are trapped in the network.
We wish to thank our many colleagues at the Royal Free Hospital and in Birmingham who have commented on the various chapters and made helpful suggestions for improvements. We are also indebted to our publishers, Wiley - Blackwell, and particularly to Rebecca Huxley who has provided tremendous skills throughout the assembly of this new edition, and Jane Fellows who has expertly drawn all the line diagrams.
A.V. Hoff brand and P.A.H. Moss
November 2010
Preface to the First Edition
The major changes that have occurred in all fields of medicine over the last decade have been accompanied by an increased understanding of the biochemical, physiological and immunological processes involved in normal blood cell formation and function and the disturbances that may occur in different diseases. At the same time, the range of treatment available for patients with diseases of the blood and blood-forming organs has widened and improved substantially as understanding of the disease processes has increased and new drugs and means of support care have been introduced.
We hope the present book will enable the medical student of the 1980s to grasp the essential features of modern clinical and laboratory haematology and to achieve an understanding of how many of the manifestations of blood diseases can be explained with this new knowledge of the disease processes.
We would like to thank many colleagues and assistants who have helped with the preparation of the book. In particular, Dr H.G. Prentice cared for the patients whose haematological responses are illustrated in Figs 5.3 and 7.8 and Dr J. McLaughlin supplied Fig. 8.6. Dr S. Knowles reviewed critically the final manuscript and made many helpful suggestions. Any remaining errors are, however, our own. We also thank Mr J.B. Irwin and R.W. McPhee who drew many excellent diagrams, Mr Cedric Gilson for expert photomicrography, Mrs T. Charalambos, Mrs B. Elliot, Mrs M. Evans and Miss J. Allaway for typing the manuscript, and Mr Tony Russell of Blackwell Scientific Publications for his invaluable help and patience.
AVH, JEP
1980
How to get the best out of your textbook
Welcome to the new edition of Essential Haematology. Over the next two pages you will be shown how to make the most of the learning features included in the textbook
An interactive textbook
For the first time, your textbook gives you free access to a Wiley Desktop Edition – a digital, interactive version of this textbook. You can view your book on a PC, Mac, laptop and Apple mobile device, and it allows you to:

Search: Save time by finding terms and topics instantly in your book, your notes, even your whole library (once you’ve downloaded more textbooks)
Note and Highlight: Colour code highlights and make digital notes right in the text so you can find them quickly and easily
Organize: Keep books, notes and class materials organized in folders inside the application
Share: Exchange notes and highlights with friends, classmates and study groups
Upgrade: Your textbook can be transferred when you need to change or upgrade your computer or device
Link: Link directly from the page of your interactive textbook to all of the material contained on the companion website
Simply find your unique Wiley Desktop Edition product code on the inside front cover of this textbook and carefully scratch away the top coating on the label, then visit
/ to get started
Full support is available at /
A companion website
Your textbook is also accompanied by a FREE companion website that contains:

Log on to to find out more
Features contained within your textbook
Every chapter has its own chapter-opening page that offers a list of key topics contained within the chapter

Throughout your textbook you will find a series of icons outlining the learning features in the book:

Your textbook is full of useful photographs, illustrations and tables. The Desktop Edition version of your textbook will allow you to copy and paste any photograph or illustration into assignments, presentations and your own notes. The photographs and illustrations are also available to download from the companion website
Every chapter ends with a chapter summary which can be used for both study and revision purposes

We hope you enjoy using your new textbook. Good luck with your studies!
CHAPTER 1
Haemopoiesis
Key topics
This first chapter is concerned with the general aspects of blood cell formation (haemopoiesis). The processes that regulate haemopoiesis and the early stages of formation of red cells (erythropoiesis), granulocytes and monocytes (myelopoiesis) and platelets (thrombopoiesis) are also discussed.
Site of haemopoiesis
In the first few weeks of gestation the yolk sac is the main site of haemopoiesis. However, definitive haemopoiesis derives from a population of stem cells first observed on the dorsal aorta termed the AGM (aorta-gonads-mesonephros) region. These common precursors of endothelial and haemopoietic cells (haemangioblasts) are believed to seed the liver, spleen and bone marrow and from 6 weeks until 6–7 months of fetal life the liver and spleen are the major haemopoietic organs and continue to produce blood cells until about 2 weeks after birth (; see b). The bone marrow is the most important site from 6 to 7 months of fetal life. During normal childhood and adult life the marrow is the only source of new blood cells. The developing cells are situated outside the bone marrow sinuses; mature cells are released into the sinus spaces, the marrow microcirculation and so into the general circulation.
Sites of haemopoiesis.
| Fetus | 0–2 months (yolk sac) |
| 2–7 months (liver, spleen) | |
| 5–9 months (bone marrow) | |
| Infants | Bone marrow (practically all bones) |
| Adults | Vertebrae, ribs, sternum, skull, sacrum and pelvis, proximal ends of femur |
In infancy all the bone marrow is haemopoietic but during childhood there is progressive fatty replacement of marrow throughout the long bones so that in adult life haemopoietic marrow is confined to the central skeleton and proximal ends of the femurs and humeri ( ). Even in these haemopoietic areas, approximately 50% of the marrow consists of fat (). The remaining fatty marrow is capable of reversion to haemopoiesis and in many diseases there is also expansion of haemopoiesis down the long bones. Moreover, the liver and spleen can resume their fetal haemopoietic role (‘extramedullary haemopoiesis’).
A normal bone marrow trephine biopsy (posterior iliac crest). Haematoxylin and eosin stain; approximately 50% of the intertrabecular tissue is haemopoietic tissue and 50% is fat.
Haemopoietic stem and progenitor cells
Haemopoiesis starts with a pluripotential stem cell that can self-renew but also give rise to the separate cell lineages. These cells are able to repopulate a bone marrow from which all stem cells have been eliminated by lethal irradiation or chemotherapy. This haemopoietic stem cell is rare, perhaps 1 in every 20 million nucleated cells in bone marrow. Although its exact phenotype is unknown, on immunological testing it is CD34+ CD38− and negative for lineage markers (Lin−) and has the appearance of a small or medium-sized lymphocyte (see ). The cells reside in specialized ‘niches’. Cell differentiation occurs from the stem cell via committed haemopoietic progenitors which are restricted in their developmental potential (). The existence of the separate progenitor cells can be demonstrated by in vitro culture techniques. Very early progenitors are assayed by culture on bone marrow stroma as long-term culture initiating cells whereas late progenitors are generally assayed in semi-solid media. An example is the earliest detectable mixed myeloid precursor which gives rise to granulocytes, erythrocytes, monocytes and megakaryocytes and is termed CFU (colony-forming unit)-GEMM (). The bone marrow is also the primary site of origin of lymphocytes (see Chapter 9) which differentiate from a common lymphoid precursor.
Diagrammatic representation of the bone marrow pluripotent stem cell and the cell lines that arise from it. Various progenitor cells can be identified by culture in semi-solid medium by the type of colony they form. It is possible that an erythroid/megakaryocytic progenitor may be formed before the common lymphoid progenitor diverges from the mixed granulocytic/monocyte/eosinophil myeloid progenitor. Baso, basophil; BFU, burst - forming unit; CFU, colony-forming unit; E, erythroid; Eo, eosinophil; GEMM, granulocyte, erythroid, monocyte and megakaryocyte; GM, granulocyte, monocyte; Meg, megakaryocyte; NK, natural killer.
The stem cell has the capability for self-renewal () so that marrow cellularity remains constant in a normal healthy steady state. There is considerable amplification in the system: one stem cell is capable of producing about 106 mature blood cells after 20 cell divisions (). The precursor cells are, however, capable of responding to haemopoietic growth factors with increased production of one or other cell line when the need arises. The development of the mature cells (red cells, granulocytes, monocytes, megakaryocytes and lymphocytes) is considered further in other sections of this book.
(a) Bone marrow cells are increasingly differentiated and lose the capacity for self - renewal as they mature. (b) A single stem cell gives rise, after multiple cell divisions (shown by vertical lines), to > 106 mature cells.
Bone marrow stroma
The bone marrow forms a suitable environment for stem cell survival, self-renewal and formation of differentiated progenitor cells. It is composed of stromal cells and a microvascular network (). The stromal cells include adipocytes, fibroblasts, osteoblasts, endothelial cells and macrophages and they secrete extracellular molecules such as collagen, glycoproteins (fibronectin and thrombospondin) and glycosaminoglycans (hyaluronic acid and chondroitin derivatives) to form an extracellular matrix. In addition, stromal cells secrete several growth factors necessary for stem cell survival.
Haemopoiesis occurs in a suitable microenvironment (‘niche’) provided by a stromal matrix on which stem cells grow and divide. There are specific recognition and adhesion sites (see p. 13); extracellular glycoproteins and other compounds are involved in the binding.
Mesenchymal stem cells , also called multipotent mesenchymal stromal cells or adherent stromal cells, are critical in stromal cell formation. Together with osteoblasts they form niches and provide the growth factors, adhesion molecules and cytokines which support stem cells, e.g. the protein jagged, on stromal cells binds to a receptor NOTCH1 on stem cells which then becomes a transcription factor involved in the cell cycle.
Stem cells are able to traffic around the body and are found in peripheral blood in low numbers. In order to exit the bone marrow, cells must cross the blood vessel endothelium and this process of mobilization is enhanced by administration of growth factors such as granulocyte colony - stimulating factor (G-CSF) (see p. 6 ). The reverse process of stem cell homing appears to depend on a chemokine gradient in which the stromal-derived factor 1 (SDF-1) is critical. Several critical interactions maintain stem cell viability and production in the stroma including stem cell factor (SCF) and jagged proteins expressed on stroma and their respective receptors KIT and NOTCH expressed on stem cell.
Tissue-specific stem cells
Stem cells are present in many different organs. These are pluripotent and can generate various types of tissue, e.g. epithelial cells, nerve cells (). Studies in patients and animals who have received haemopoietic stem cell transplants (see Chapter 23) have suggested that donor haemopoietic cells may contribute to tissues such as neurons, liver and muscle. The contribution of adult donor haemopoietic cells to non-haemopoietic tissues is at most very small. The persistence of pluripotential stem cells in postnatal life, the presence of mesenchymal stem cells in bone marrow and fusion of transplanted cells with host cells have all been proposed to explain many of the findings suggesting stem cell ‘plasticity’.
(a) Cells from the inner cell mass of the blastocyst in the early embryo are able to generate all the tissues of the body and are known as totipotent. (b) Specialized adult stem cells of the bone marrow, nervous tissue, epithelial and other tissues give rise to differentiated cells of the same tissue.
The regulation of haemopoiesis
Haemopoiesis starts with stem cell division in which one cell replaces the stem cell (self-renewal) and the other is committed to differentiation. These early committed progenitors express low levels of transcription factors that may commit them to discrete cell lineages. Which cell lineage is selected for differentiation may depend both on chance and on the external signals received by progenitor cells. Several transcription factors (see p. 13 ) regulate survival of stem cells (e.g. SCL, GATA-2, NOTCH-1) whereas others are involved in differentiation along the major cell lineages. For instance, PU.1 and the CEBP family commit cells to the myeloid lineage whereas GATA-1 and FOG-1 have an essential roles in erythropoietic and megakaryocytic differentiation.
Haemopoietic growth factors
The haemopoietic growth factors are glycoprotein hormones that regulate the proliferation and differentiation of haemopoietic progenitor cells and the function of mature blood cells. They may act locally at the site where they are produced by cell–cell contact or circulate in plasma. They also bind to the extracellular matrix to form niches to which stem and progenitor cells adhere. The growth factors may cause cell proliferation but can also stimulate differentiation, maturation, prevent apoptosis and affect the function of mature cells ().
Growth factors may stimulate proliferation of early bone marrow cells, direct differentiation to one or other cell type, stimulate cell maturation, suppress apoptosis or affect the function of mature non - dividing cells, as illustrated here for granulocyte colony-stimulating factor (G-CSF) for an early myeloid progenitor and a neutrophil.
They share a number of common properties () and act at different stages of haemopoiesis ( ; ). Stromal cells are the major source of growth factors except for erythropoietin, 90% of which is synthesized in the kidney, and thrombopoietin, made largely in the liver. An important feature of growth factor action is that two or more factors may synergize in stimulating a particular cell to proliferate or differentiate. Moreover, the action of one growth factor on a cell may stimulate production of another growth factor or growth factor receptor. SCF and FLT ligand (FLT-L) act locally on the pluripotential stem cells and on early myeloid and lymphoid progenitors (). Interleukin-3 (IL-3) and granuloctye–macrophage colony-stimulating factor (GM-CSF) are multipotential growth factors with overlapping activities. G-CSF and thrombopoietin enhance the effects of SCF, FLT-L, IL-3 and GM-CSF on survival and differentiation of the early haemopoietic cells.
A diagram of the role of growth factors in normal haemopoiesis. Multiple growth factors act on the earlier marrow stem and progenitor cells. EPO, erythropoietin; PSC, pluripotential stem cell; SCF, stem cell factor; TPO, thrombopoietin. For other abbreviations see .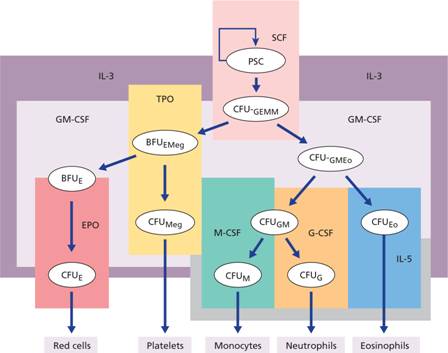
General characteristics of myeloid and lymphoid growth factors.
| Glycoproteins that act at very low concentrations |
| Act hierarchically |
| Usually produced by many cell types |
| Usually affect more than one lineage |
| Usually active on stem/progenitor cells and on functional end cells |
| Usually show synergistic or additive interactions with other growth factors |
| Often act on the neoplastic equivalent of a normal cell |
| Multiple actions: proliferation, differentiation, maturation, functional activation, prevention of apoptosis of progenitor cells |
Haemopoietic growth factors.
| Act on stromal cells |
| IL-1 |
| TNF |
| Act on pluripotential stem cells |
| SCF |
| FLT3-L |
| VEGF |
| Act on multipotential progenitor cells |
| IL-3 |
| GM-CSF |
| IL-6 |
| G-CSF |
| Thrombopoietin |
| Act on committed progenitor cells |
| G-CSF * |
| M-CSF |
| IL-5 (eosinophil-CSF) |
| Erythropoietin |
| Thrombopoietin* |
CSF, colony-stimulating factor; FLT3-L, FLT3 ligand;
G-CSF, granulocyte colony-stimulating factor; GM-CSF,
granulocyte–macrophage colony - stimulating factor; IL,
interleukin; M-CSF, macrophage colony-stimulating factor;
SCF, stem cell factor; TNF, tumour necrosis factor; VEGF,
vascular endothelial growth factor.
* These also act synergistically with early acting factors on pluripotential progenitors.
These factors maintain a pool of haemopoietic stem and progenitor cells on which later acting factors erythropoietin, G-CSF, macrophage colony-stimulating factor (M-CSF), IL-5 and thrombopoietin act to increase production of one or other cell lineage in response to the body’s need. Granulocyte and monocyte formation, for example, can be stimulated by infection or inflammation through release of IL-1 and tumour necrosis factor (TNF) which then stimulate stromal cells to produce growth factors in an interacting network (see ). In contrast, cytokines such as transforming growth factor-β (TGF-β) and γ-interferon (IFN-γ) can exert a negative effect on haemopoiesis and may have a role in the development of aplastic anaemia (see p. 290).
Growth factor receptors and signal transduction
The biological effects of growth factors are mediated through specific receptors on target cells. Many receptors (e.g. erythropoietin (epo) receptor (R), GMCSF-R) are from the haematopoietin receptor superfamily which dimerize after binding their ligand.
Dimerization of the receptor leads to activation of a complex series of intracellular signal transduction pathways of which the three major ones are the JAK/STAT, the mitogen-activated protein (MAP) kinase and the phosphatidylinositol 3 (PI3) kinase pathways (; see ). The Janus-associated kinase (JAK) proteins are a family of four tyrosine-specific protein kinases that associate with the intracellular domains of the growth factor receptors (). A growth factor molecule binds simultaneously to the extracellular domains of two or three receptor molecules, resulting in their aggregation. Receptor aggregation induces activation of the JAKs which now phosphorylate members of the signal transducer and activator of transcription (STAT) family of transcription factors. This results in their dimerization and translocation from the cell cytoplasm across the nuclear membrane to the cell nucleus. Within the nucleus STAT dimers activate transcription of specific genes. A model for control of gene expression by a transcription factor is shown in Fig. . The clinical importance of this pathway is revealed by the finding of an activating mutation of the JAK2 gene as the cause of polycythaemia rubra vera (see p. 201).
Control of haemopoiesis by growth factors. The factors act on cells expressing the corresponding receptors. Binding of a growth factor to its receptor activates the JAK/STAT, MAPK and phosphatidyl-inositol 3-kinase (PI3K) pathways (see ) which leads to transcriptional activation of specific genes. E2F is a transcription factor needed for cell transition from G1 to S phase. E2F is inhibited by the tumour suppressor gene Rb (retinoblastoma) which can be indirectly activated by p53. The synthesis and degradation of different cyclins () stimulates the cell to pass through the different phases of the cell cycle. The growth factors may also suppress apoptosis by activating AKT (protein kinase B).
Model for control of gene expression by a transcription factor. The DNA - binding domain of a transcription factor binds a specific enhancer sequence adjacent to a structural gene. The transactivation domain then binds a molecule of RNA polymerase, thus augmenting its binding to the TATA box. The RNA polymerase now initiates transcription of the structural gene to form mRNA. Translation of the mRNA by the ribosomes generates the protein encoded by the gene.
JAK can also activate the MAPK pathway which is regulated by Ras and controls proliferation. PI3 kinases phophorylate inositol lipids which have a wide range of downstream effects including activation of AKT leading to block of apoptosis and other actions (; see ). Different domains of the intracellular receptor protein may signal for the different processes (e.g. proliferation or suppression of apoptosis) mediated by growth factors.
A second smaller group of growth factors, including SCF, FLT-3L and M-CSF (), bind to receptors that have an extracellular immunoglobulin-like domain linked via a transmembrane bridge to a cytoplasmic tyrosine kinase domain. Growth factor binding results in dimerization of these receptors and consequent activation of the tyrosine kinase domain. Phosphorylation of tyrosine residues in the receptor itself generates binding sites for signalling proteins which initiate complex cascades of biochemical events resulting in changes in gene expression, cell proliferation and prevention of apoptosis.
The cell cycle
The cell division cycle, generally known simply as the cell cycle, is a complex process that lies at the heart of haemopoiesis. Dysregulation of cell proliferation is also the key to the development of malignant disease. The duration of the cell cycle is variable between different tissues but the basic principles remain constant. The cycle is divided into the mitotic phase (M phase), during which the cell physically divides, and interphase during which the chromosomes are duplicated and cell growth occurs prior to division (). The M phase is further partitioned into classical mitosis in which nuclear division is accomplished, and cytokinesis in which cell fission occurs.
(a) The stages of the cell cycle. Progression through cell cycle is regulated by specific combinations of cyclin-dependent protein kinases (Cdk) and cyclin proteins. The synthesis and degradation of different cyclins stimulates the cell to pass through the different phases of the cell cycle. (b) Relationship between the DNA content of a cell expressed in arbitrary units as 2c increasing to 4c and its position in the cell cycle. (Adapted from Wickramasinghe S.N. (1975) Human Bone Marrow, Blackwell Scientific, Oxford, p. 13.)
Interphase is divided into three main stages: a G1 phase in which the cell begins to commit to replication, an S phase during which DNA content doubles () and the chromosomes replicate and the G2 phase in which the cell organelles are copied and cytoplasmic volume is increased. If cells rest prior to division they enter a G0 state where they can remain for long periods of time. The number of cells at each stage of the cell cycle can be assessed by exposing cells to a chemical or radiolabel that gets incorporated into newly generated DNA or by flow cytometry.
The cell cycle is controlled by two checkpoints which act as brakes to coordinate the division process at the end of the G1 and G2 phases. Two major classes of molecules control these checkpoints, cyclin-dependent protein kinases (Cdk) which phosophorylate downstream protein targets and cyclins which bind to Cdks and regulate their activity. An example of the importance of these systems is demonstrated by mantle cell lymphoma which results from the constitutive activation of cyclin D1 as a result of a chromosomal translocation (see p. 267).
Apoptosis
Apoptosis (programmed cell death) is a regulated process of physiological cell death in which individual cells are triggered to activate intracellular proteins that lead to the death of the cell. Morphologically it is characterized by cell shrinkage, condensation of the nuclear chromatin, fragmentation of the nucleus and cleavage of DNA at internucleosomal sites. It is an important process for maintaining tissue homeostasis in haemopoiesis and lymphocyte development.
Apoptosis results from the action of intracellular cysteine proteases called caspases which are activated following cleavage and lead to endonuclease digestion of DNA and disintegration of the cell skeleton (). There are two major pathways by which caspases can be activated. The first is by signalling through membrane proteins such as Fas or TNF receptor via their intracellular death domain. An example of this mechanism is shown by activated cytotoxic T cells expressing FAS ligand which induce apoptosis in target cells. The second pathway is via the release of cytochrome c from mitochondria. Cytochrome c binds to APAF-1 which then activates caspases. DNA damage induced by irradiation or chemotherapy may act through this pathway. The protein p53 has an important role in sensing DNA damage. It activates apoptosis by raising the cell level of BAX which then increases cytochrome c release (). P53 also shuts down the cell cycle to stop the damaged cell from dividing (). The cellular level of p53 is rigidly controlled by a second protein MDM2. Following death, apoptotic cells display molecules that lead to their ingestion by macrophages.
Representation of apoptosis. Apoptosis is initiated via two main stimuli: (i) signalling through cell membrane receptors such as FAS or tumour necrosis factor (TNF) receptor; or (ii) release of cytochrome c from mitochondria. Membrane receptors signal apoptosis through an intracellular death domain leading to activation of caspases which digest DNA. Cytochrome c binds to the cytoplasmic protein Apaf - 1 leading to activation of caspases. The intracellular ratio of pro - apoptotic (e.g. BAX) or anti - apoptotic (e.g. BCL - 2) members of the BCL - 2 family may influence mitochondrial cytochrome c release. Growth factors raise the level of BCL - 2 inhibiting cytochrome c release whereas DNA damage, by activating p53, raises the level of BAX which enhances cytochrome c release.
As well as molecules that mediate apoptosis there are several intracellular proteins that protect cells from apoptosis. The best characterized example is BCL-2. BCL-2 is the prototype of a family of related proteins, some of which are anti-apoptotic and some, like BAX, pro-apoptotic. The intracellular ratio of BAX and BCL-2 determines the relative susceptibility of cells to apoptosis (e.g. determines the lifespan of platelets) and may act through regulation of cytochrome c release from mitochondria.
Many of the genetic changes associated with malignant disease lead to a reduced rate of apoptosis and hence prolonged cell survival. The clearest example is the translocation of the BCL-2 gene to the immunoglobulin heavy chain locus in the t(14; 18) translocation in follicular lymphoma. Overexpression of the BCL-2 protein makes the malignant B cells less susceptible to apoptosis. Apoptosis is the normal fate for most B cells undergoing selection in the lymphoid germinal centres.
Several translocations leading to the generation of fusion proteins such as t(9; 22), t(1; 14) and t(15; 17) also result in inhibition of apoptosis (see Chapter 11). In addition, genes encoding proteins that are involved in mediating apoptosis following DNA damage, such as p53 and ATM, are also frequently mutated and therefore inactivated in haemopoietic malignancies.
Necrosis is death of cells and adjacent cells due to ischemia, chemical trauma or hyperthermia. The cells swell, the plasma membrane loses integrity. There is usually an inflammatory infiltrate in response to spillage of cell contents. Autophagy is the digestion of cell organelles by lysosomes. It may be involved in cell death but in some situations also in maintaining cell survival by recycling nutrients.
Transcription factors
Transcription factors regulate gene expression by controlling the transcription of specific genes or gene families. Typically, they contain at least two domains: a DNA-binding domain such as a leucine zipper or helix-loop-helix motif which binds to a specific DNA sequence, and an activation domain which contributes to assembly of the transcription complex at a gene promoter (). Mutation, deletion or translocation of transcription factors underlie many cases of haematological neoplasms.
Adhesion molecules
A large family of glycoprotein molecules termed adhesion molecules mediate the attachment of marrow precursors, leucocytes and platelets to various components of the extracellular matrix, to endothelium, to other surfaces and to each other. The adhesion molecules on the surface of leucocytes are termed receptors and these interact with molecules (termed ligands) on the surface of potential target cells. Three main families exist:
1 Immunoglobulin superfamily This includes receptors that react with antigens (the T-cell receptors and the immunoglobulins) and antigen-independent surface adhesion molecules.
2 Selectins These are mainly involved in leucocyte and platelet adhesion to endothelium during inflammation and coagulation.
3 Integrins These are involved in cell adhesion to extracellular matrix (e.g. to collagen in wound healing and in leucocyte and platelet adhesion).
The adhesion molecules are thus important in the development and maintenance of inflammatory and immune responses, and in platelet–vessel wall and leucocyte–vessel wall interactions. Expression of adhesion molecules can be modifed by extracellular and intracellular factors and this alteration of expression may be quantitative or functional. IL-1, TNF, IFN-γ , T-cell activation, adhesion to extracellular proteins and viral infection may all up-regulate expression of these molecules.
The pattern of expression of adhesion molecules on tumour cells may determine their mode of spread and tissue localization (e.g. the pattern of metastasis of carcinoma cells or non-Hodgkin lymphoma cells into a follicular or diffuse pattern). The adhesion molecules may also determine whether or not cells circulate in the bloodstream or remain fixed in tissues. They may also partly determine whether or not tumour cells are susceptible to the body’s immune defences.
SUMMARY
 Now visit to test yourself on this chapter.
Now visit to test yourself on this chapter.