

Table of Contents
Related Titles
Title Page
Copyright
Preface
List of Contributors
Introduction
Chapter 1: The Significance of Heterocycles for Pharmaceuticalsand Agrochemicals
1.1 Introduction
1.2 Heterocycles as Framework of Biologically Active Compounds
1.3 Fine-Tuning the Physicochemical Properties with Heterocycles
1.4 Heterocycles as Prodrugs
1.5 Heterocycles as Peptidomimetics
1.6 Heterocycles as Isosteric Replacement of Functional Groups
1.7 Heterocycles as Isosteric Replacement of Alicyclic Rings
1.8 Heterocycles as Isosteric Replacement of other Heterocyclic Rings
References
Part I: Herbicides
Chapter 2: Triazine Herbicides
2.1 Introduction
2.2 History
2.3 Synthesis
2.4 Mode of Action
2.5 Biological Activity
2.6 Structure–Activity Relationships
References
Chapter 3: Pyrimidinyl and Triazinylsulfonylurea Herbicides
3.1 Introduction
3.2 History
3.3 Synthesis
3.4 Mode of Action
3.5 Biological Activity
3.6 Structure–Activity Relationship
References
Chapter 4: Acetohydroxyacid Synthase Inhibiting Triazolopyrimidine Herbicides
4.1 Introduction
4.2 History
4.3 Synthesis
4.4 Mode of Action
4.5 Biological Activity
4.6 Structure–Activity Relationship
Chapter 5: HPPD-Inhibiting Benzoylpyrazole Herbicides
5.1 Introduction
5.2 History
5.3 Synthesis
5.4 Mode of Action
5.5 Biological Activity
5.6 Structure–Activity Relationship
References
Chapter 6: Pyridyloxyphenoxypropionate Herbicides: Inhibitors of Acetyl-CoA Carboxylase
6.1 Introduction
6.2 History
6.3 Synthesis
6.4 Mode of Action
6.5 Biological Activity
6.6 Structure–Activity Relationships
References
Chapter 7: Imidazolinone Herbicides
7.1 Introduction
7.2 History
7.3 Synthesis
7.4 Mode of Action
7.5 Biological Activity
7.6 Structure–Activity Relationship
References
Chapter 8: Protoporphyrinogen-IX-Oxidase-Inhibiting Uracil Herbicides
8.1 Introduction
8.2 History
8.3 Synthesis
8.4 Mode of Action
8.5 Biological Activity
8.6 Structure–Activity Relationship
References
Part II: Fungicides
Chapter 9: Benzimidazole Fungicides
9.1 Introduction
9.2 History
9.3 Synthesis
9.4 Mode of Action
9.5 Biological Activity
9.6 Structure–Activity Relationship
References
Chapter 10: Morpholine Fungicides for the Treatment of Powdery Mildew
10.1 Introduction
10.2 History
10.3 Synthesis
10.4 Mode of Action
10.5 Biological Activity
10.6 Structure–Activity Relationship
References
Chapter 11: Sterol Biosynthesis Inhibiting Triazole Fungicides
11.1 Introduction
11.2 History
11.3 Synthesis
11.4 Mode of Action
11.5 Biological Activity
11.6 Structure–Activity Relationship
References
Chapter 12: Methionine Biosynthesis-Inhibiting Anilinopyrimidine Fungicides
12.1 Introduction
12.2 History
12.3 Synthesis
12.4 Mode of Action
12.5 Biological Activity
12.6 Structure–Activity Relationship
References
Chapter 13: Phenylpyrrole Fungicides
13.1 Introduction
13.2 History
13.3 Synthesis
13.4 Mode of Action
13.5 Biological Activity
13.6 Structure–Activity Relationship
References
Chapter 14: Broad-Spectrum Fungicidally Active Pyrimidinyldioxy Strobilurins Inhibiting the Respiratory Chain
14.1 Introduction
14.2 History
14.3 Synthesis
14.4 Mode of Action
14.5 Biological Activity
14.6 Structure–Activity Relationship
References
Chapter 15: Pyrazole Carboxamide Fungicides Inhibiting Succinate Dehydrogenase
15.1 Introduction
15.2 History
15.3 Synthesis
15.4 Mode of Action
15.5 Biological Activity
15.6 Structure–Activity Relationships
Acknowledgements
References
Part III: Insecticides
Chapter 16: Avermectin Insecticides and Acaricides
16.1 Introduction
16.2 History
16.3 Synthesis
16.4 Mode of Action
16.5 Biological Activity
16.6 Structure–Activity Relationship
References
Chapter 17: Pyridine and Thiazole-Containing Insecticides as Potent Agonists on Insect Nicotinic Acetylcholine Receptors
17.1 Introduction
17.2 History
17.3 Synthesis
17.4 Mode of Action
17.5 Biological Activity
17.6 Structure–Activity Relationship
References
Chapter 18: Pyrazole and Pyrimidine Acaricides and Insecticides Acting as Inhibitors of Mitochondrial Electron Transport at Complex I
18.1 Introduction
18.2 History
18.3 Synthesis
18.4 Mode of Action
18.5 Biological Activity
18.6 Structure–Activity Relationship
References
Chapter 19: Phenylpyrazole-Containing Fiprole Insecticides
19.1 Introduction
19.2 History
19.3 Synthesis
19.4 Mode of Action
19.5 Biological Activity
19.6 Structure–Activity Relationship
References
Chapter 20: Pyrazolylpyridine Activators of the Insect Ryanodine Receptor
20.1 Introduction
20.2 History
20.3 Synthesis
20.4 Mode of Action
20.5 Biological Activity
20.6 Structure–Activity Relationships
References
Chapter 21: Tetronic Acid Insecticides and Acaricides Inhibiting Acetyl-CoA Carboxylase
21.1 Introduction
21.2 History
21.3 Synthesis
21.4 Mode of Action
21.5 Biological Activity
21.6 Structure–Activity Relationship
References
Index
Related Titles
Jeschke, P., Krämer, W., Schirmer, U.,
Witschel, M. (eds.)
Modern Methods in Crop Protection Research
2012
Hardcover
ISBN: 978-3-527-33175-8
Dinges, J., Lamberth, C. (eds.)
Bioactive Heterocyclic Compound Classes
Pharmaceuticals
2012
Hardcover
ISBN: 978-3-527-33395-0
Krämer, W., Schirmer, U., Jeschke, P.,
Witschel, M. (eds.)
Modern Crop Protection Compounds
2012
Hardcover
ISBN: 978-3-527-32965-6
Majumdar, K. C., Chattopadhyay, S. K. (eds.)
Heterocycles in Natural Product Synthesis
2011
Hardcover
ISBN: 978-3-527-32706-5
Royer, J. (ed.)
Asymmetric Synthesis of Nitrogen Heterocycles
2009
Hardcover
ISBN: 978-3-527-32036-3

The Editors
Dr. Clemens Lamberth
Syngenta Crop Protection AG
Research Chemistry
Schaffhauserstr. 101
4332 Stein
Schweiz
Dr. Jürgen Dinges
Abbott Laboratories
Global Pharmaceutical R&D
200, Abbott Park Road
Abbott Park, IL 60064-6217
USA
All books published by Wiley-VCH are carefully produced. Nevertheless, authors, editors, and publisher do not warrant the information contained in these books, including this book, to be free of errors. Readers are advised to keep in mind that statements, data, illustrations, procedural details or other items may inadvertently be inaccurate.
Library of Congress Card No.: applied for
British Library Cataloguing-in-Publication Data
A catalogue record for this book is available from the British Library.
Bibliographic information published by the Deutsche Nationalbibliothek
The Deutsche Nationalbibliothek lists this publication in the Deutsche Nationalbibliografie; detailed bibliographic data are available on the Internet at <http://dnb.d-nb.de>.
© 2012 Wiley-VCH Verlag & Co. KGaA, Boschstr. 12, 69469 Weinheim, Germany
All rights reserved (including those of translation into other languages). No part of this book may be reproduced in any form — by photoprinting, microfilm, or any other means — nor transmitted or translated into a machine language without written permission from the publishers. Registered names, trademarks, etc. used in this book, even when not specifically marked as such, are not to be considered unprotected by law.
Bioactive Heterocyclic Compound Classes (Pharmaceuticals and Agrochemicals, 2 Volume Set)
ISBN: 978-3-527-32993-9
Print ISBN: 978-3-527-33396-7
ePDF ISBN: 978-3-527-66444-3
ePub ISBN: 978-3-527-66443-6
mobi ISBN: 978-3-527-66442-9
oBook ISBN: 978-3-527-66441-2
Preface
Approximately 70% of all the 2400 pharmaceuticals listed in the online version of “Pharmaceutical Substances” (A. Kleemann et al., Thieme) bear at least one heterocyclic ring; the latest edition of the “Pesticide Manual” (C. D. S. Tomlin, BCPC) contains a similar percentage of heterocyclic agrochemicals among its about 900 entries. This vast number of known pharmaceuticals and agrochemicals makes the field of commercialized active ingredients an unmanageable jungle. Only specialists are able to understand the connectivities of these active ingredients, many of which are heterocycles.
Therefore, we decided to put this book together, which tries to show the relationship of those heterocyclic active ingredients, which belong together, forming a biologically active heterocylic chemistry class. According to our definition, such a heterocyclic family is built from at least three compounds that fulfill the following conditions: (i) same heterocyclic scaffold, (ii) same mode of action, and (iii) similar substitution pattern.
Although the strength of this concept is that for the first time the members of the most important heterocyclic active ingredient families, their historical background, chemical syntheses, biochemical modes of action, and biological activities are discussed in detail, there are also some limitations. For instance, there are some heterocyclic families of drugs or crop protection agents, such as the analgesic COX-2 inhibitors celecoxib, valdecoxib, and rofecoxib or the dicarboxamide fungicides vinclozolin, iprodione, and procymidone, which are closely related by structure and possess the same mode of action, but bear different heterocyclic scaffolds and therefore could not be considered.
We would like to thank the authors of the 40 chapters of this book, all of them experts in their field, for spending their scarce time summarizing their area of interest. They all agreed to write the chapters according to the same outline: (i) introduction, (ii) history, (iii) synthesis, (iv) mode of action, and (v) structure–activity relationship. Only the agrochemical chapters possess an additional section “biological activity,” mainly describing the target spectrum of the active ingredients. This book would definitely not exist without your engagement!
Furthermore, we also would like to thank Anne Brennführer and Stefanie Volk of Wiley-VCH, who from the beginning guided us very efficiently through all different phases of this exciting project.
The introductory chapter about “The significance of heterocycles for pharmaceuticals and agrochemicals” tries to explain the different roles of heterocyclic scaffolds in active ingredients, e.g. as framework of biologically active substances, as prodrugs, as tool for fine-tuning the physicochemical properties, as isosteric replacements of functional groups, alicyclic rings or other heterocyclic rings. As this is demonstrated at the hand of many prominent and characteristical examples of pharmaceuticals as well as of agrochemicals, also pointing out the many similarities, but also some differences between the two big classes of active ingredients, we decided to put this chapter in front of both volumes.
Although currently living in two different continents, both of us enjoyed exactly the same excellent education, a Ph.D. in organic chemistry from the Technical University at Darmstadt, Germany, and a subsequent postdoctoral fellowship at the chemistry department of the University of California at Berkeley. We are very grateful to our teachers, mentors, and research advisors at both universities, who built the foundation for our successful work in the research departments of the agrochemical and pharmaceutical industry.
Finally, we are deeply indebted to our wives Annette and Petra, who continuously supported us, as always, and tolerated that we spent many hours of our spare time, which should have belonged to our families, working on this book. You really made this possible!
Clemens Lamberth
Switzerland
Jürgen Dinges
USA
List of Contributors
Introduction
1
The Significance of Heterocycles for Pharmaceuticalsand Agrochemicals*
Heterocycles, their preparation, transformation, and properties, are undoubtedly a cornerstone of organic chemistry. Several books not only on heterocyclic chemistry [1–6] but also on some special aspects, such as heterocyclic name reactions [7], heterocyclic palladium-catalyzed reactions [8], heterocyclic carbene complexes [9], and fluorinated heterocycles [10], have been published recently.
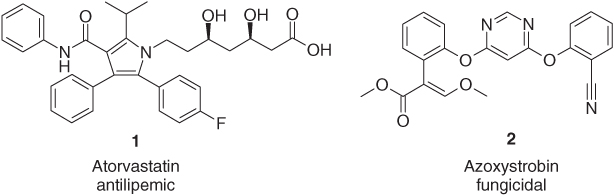
Approximately more than 70% of all pharmaceuticals and agrochemicals bear at least one heterocyclic ring. In addition, some of the biggest commercial products to date, such as the blockbuster blood cholesterol reducer atorvastatin (Lipitor®, 1) [11] for the treatment of dyslipidemia and the prevention of cardiovascular diseases and the broad-spectrum fungicide azoxystrobin (Amistar®, 2) [12], currently applied against diseases of more than 100 different crops in more than 100 different countries, belong to this huge heterocyclic group of active ingredients (Figure 1.1).
Figure 1.1 Atorvastatin (1) and azoxystrobin (2), two of the currently most successful pharmaceuticals and agrochemicals.
There are two major reasons for the tremendous value of heterocycles for the lead optimization of pharmaceuticals and agrochemicals. The heterocyclic scaffold of a drug often has a positive impact on its synthetic accessibility and its physicochemical properties, driving these values of lipophilicity and solubility toward the optimal balanced range regarding uptake and bioavailability. Furthermore, heterocycles seem to be perfect bioisosteres of other iso- or heterocyclic rings as well as of several different functional groups, in most cases, delivering through their similarity in structural shape and electronic distribution equal or even better biological efficacy [13].
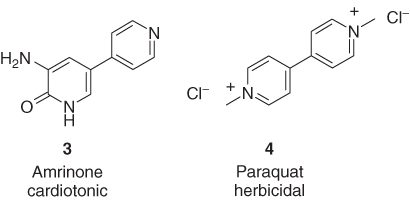
Several heterocycles possess excellent biological activity almost without bearing any substituents, which means that their heterocyclic core is definitely part of the pharmacophore. Examples of such scarcely substituted and highly active heterocycles are the two bipyridyl derivatives such as amrinone (3) [14], which is used in the treatment of congestive heart failure, and paraquat (4) [15], which is applied as a total herbicide (Figure 1.2).
Figure 1.2 The highly active bipyridyl derivatives amrinone (3) and paraquat (4), each carrying only two small substituents.
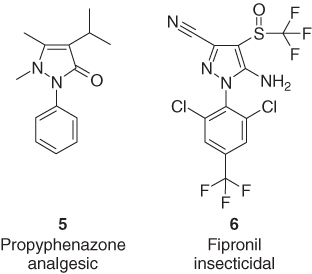
Another important role of the heterocyclic core of several pharmaceuticals and agrochemicals is that of an easily accessible scaffold, which carries the substituents that are responsible for the biological activity in the right orientation. There are several highly active per-substituted heterocycles, as demonstrated by the pyrazole derivatives propyphenazone (5) [16] and fipronil (6) [17], which are widely applied as efficient analgesic and insecticide, respectively, and synthetically available in only few steps (Figure 1.3).
Figure 1.3 The persubstituted pyrazole derivatives propyphenazone (5) and fipronil (6).
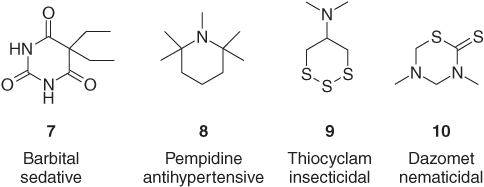
Even simple aliphatic heterocycles display astonishing biological activities. The gem-diethyl-substituted barbituric acid derivative barbital (7) has been widely applied as a sleeping aid [18]. The pentamethylated piperidine pempidine (8) is used as a ganglionic blocker [19]. The trithiane thiocyclam (9), in comparison to the marine natural product nereistoxin enlarged by one additional ring sulfur atom, has been developed as a broad-spectrum insecticide [20]. The cyclic dithiocarbamate dazomet (10) is a soil fumigant, which readily decomposes, yielding methyl isothiocyanate as principal toxicant against nematodes (Figure 1.4) [21].
Figure 1.4 The saturated bioactive heterocycles barbital (7), pempidine (8), thiocyclam (9), and dazomet (10) [18–21].
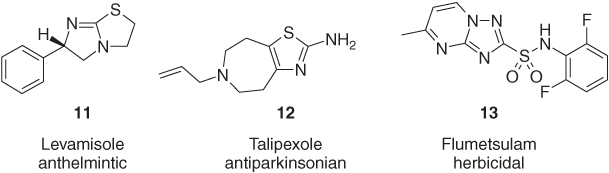
Not only monocyclic heterocycles but also annelated bicyclic ring systems are applied as pharmaceuticals and crop protection agents, regardless of whether the biheterocyclic core consists of aliphatic, aliphatic and aromatic, or purely aromatic rings. The tetrahydroimidazothiazole levamisole (11) has been used as anthelmintic and immunomodulator [22]. The dopamine agonist talipexole (12) combines a five- and seven-membered ring and has been proposed as an antiparkinsonian agent [23]. The triazolopyrimidine sulfonanilide flumetsulam (13) is used for the control of broadleaf weeds in corn and soybean (Figure 1.5) [24].
Figure 1.5 The highly active annelated bicyclic heterocycles levamisole (11), talipexole (12), and flumetsulam (13) [22–24].
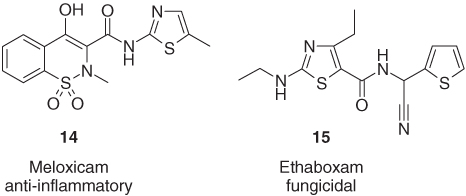
Finally, there are also several examples of active ingredients, which bear two or more heterocycles in completely different positions of the molecule. For instance, the nonsteroidal anti-inflammatory drug meloxicam (14) consists of an amide with a benzothiazine-dione acid moiety and a thiazole amine component [25]. In addition, the agrochemical fungicide ethaboxam (15) contains an amide functionality, combining a thiazole carboxylic acid with a thiophene-containing amine (Figure 1.6) [26].
Figure 1.6 Meloxicam (14) and ethaboxam (15), two active ingredients carrying heterocycles in different parts of the molecule [25, 26].
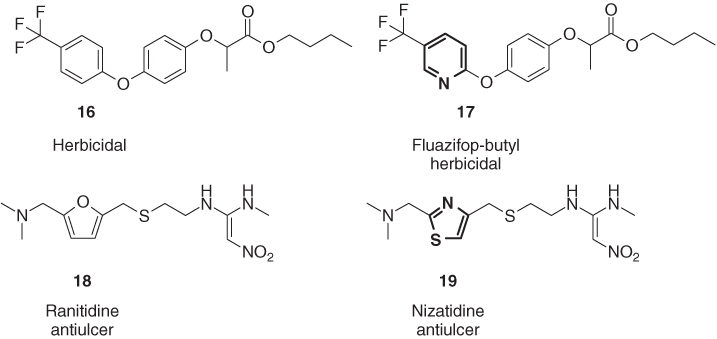
The fact that in most cases aromatic heterocycles are more polar than their isocyclic analogs is often used for the lead optimization of pharmaceuticals and agrochemicals. For example, the replacement of the 4-trifluoromethylphenyl moiety of the herbicidal lead structure 16 by a 5-CF3-pyrid-2-yl group resulting in the postemergence herbicide fluazifop-butyl (17) did not lead to any considerable enhancement of the herbicidal activity but significantly improved the ability of the target grass weeds to translocate into the plant tissue because of an optimum partition coefficient [27]. Furthermore, the replacement of the furane scaffold of the antiulcer histamine H2-receptor antagonist ranitidine (18) by a thiazole resulted in nizatidine (19), which possesses not only a considerably lower log P value than ranitidine but also a much higher human oral bioavailability (Figure 1.7) [28].
Figure 1.7 Fluazifop-butyl (17) and nizatidine (19) possess optimum physicochemical properties to transport their high intrinsic activity to the target [27, 28].
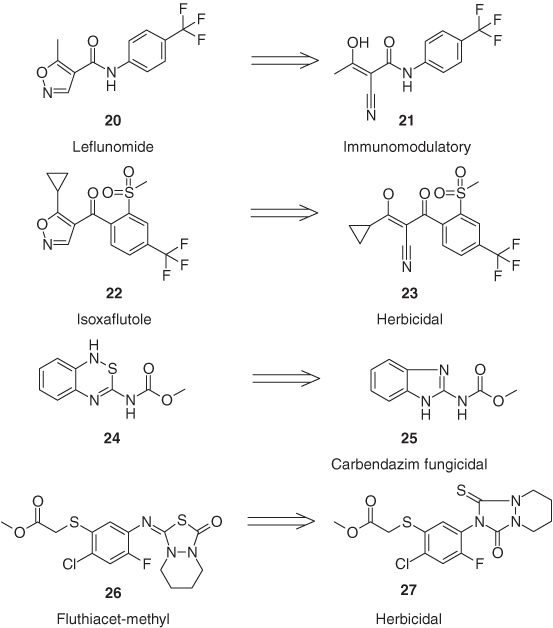
The efficacy of several heterocyclic active ingredients is based on the fact that the heterocycle is acting as a prodrug, itself being not efficacious against the target enzyme or organism but delivering the intrinsically active compound by UV light, heat, moisture, or a metabolic transformation. Leflunomide (20), for example, is a prodrug against transplant rejection, which ring-opens quantitatively in the cellular system to the hydroxypropenamide (21), which is responsible for the immunosuppressive efficacy [29]. In addition, the isoxazole ring of the herbicide isoxaflutole (22) is metabolically converted in plants and soil to the 2-cyano-1,3-diketone (23), which is a potent inhibitor of p-hydroxyphenylpyruvate dioxygenase (HPPD), one of the most important molecular targets for herbicides [30]. The fungicidal activity of the benzothiadiazine derivative 24 originates from its ability to be converted by sulfur extrusion in aqueous solutions and in plants into the benzimidazole fungicide carbendazim (25) [31]. The in vivo isomerization of fluthiacet-methyl (26) by glutathione-S-transferase leads to the urazole derivative 27, which is entirely responsible for the strong herbicidal activity (Figure 1.8) [32].
Figure 1.8 The heterocyclic prodrugs leflunomide (20), isoxaflutole (22), and fluthiacet-methyl (26) and (24).
Several different heterocyclic rings have a proven record as perfect isosteric replacement of the amide function in peptides [33]. The highly active HIV-1 protease inhibitors saquinavir (29) [34] and (30) [35] are close analogs of telinavir (28) [36], in which part of its urea function have been replaced by either a decahydroisoquinoline or a tetrazole (Figure 1.9).
Figure 1.9 Telinavir (28) and its peptidomimetics saquinavir (29) and (30).
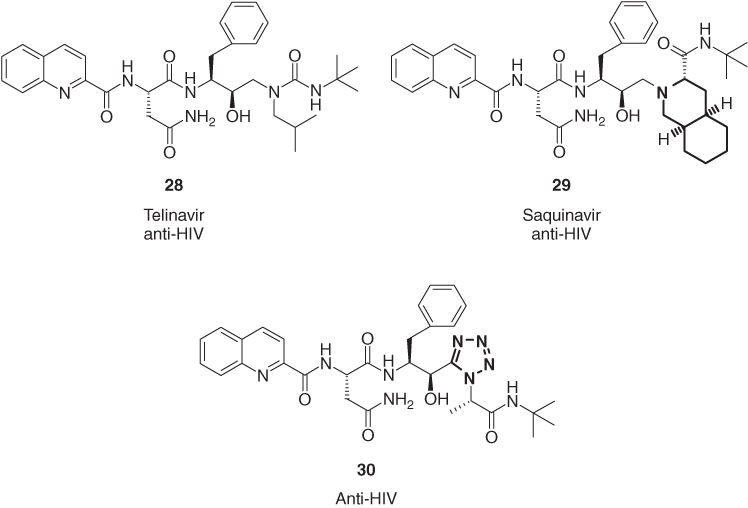
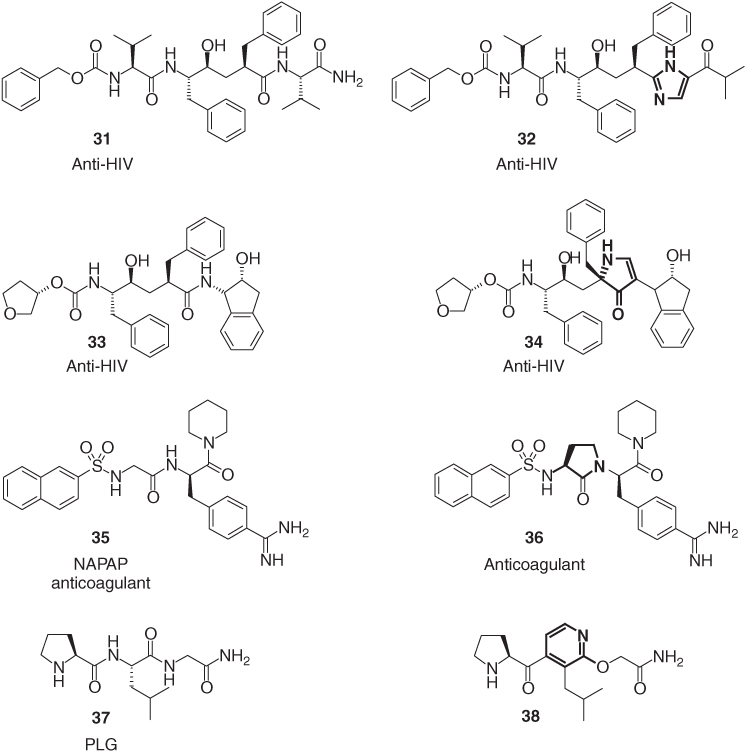
Also, other five-membered heterocycles have been applied as amide isosteres in HIV-1 protease inhibitors for the treatment of AIDS. Examples are the imidazole derivative 32 [37] and the pyrrolinone (34) [38], in which the heterocyclic ring replaces the amide function of the corresponding di- or tripeptides 31 and 33 (Figure 1.10). All four HIV-1 protease inhibitors, the peptidic drugs, as well as their heterocyclic isosteres are active in the nanomolar range. The pyrrolidinone peptidomimetic 36 is 100 times more potent than the open-chain thrombin inhibitor NAPAP (35) [39]. The pyridine-based peptidomimetic 38 is a potent analog of PLG (37) (Pro-Leu-Gly-NH2), an endogenous tripeptide found in the central nervous system, which is known to exert its pharmacological effects through the modulation of dopamine D2 receptors [40].
Figure 1.10 The heterocyclic peptidomimetics 32, 34, 36, and 38.
Further heterocycles, which have been successfully applied as amide isosteres, are pyrroles [41], thiazolidines [42], isoxazolines [43], imidazolines [44], oxazoles [45], triazoles [46], oxadiazoles [47], and benzimidazoles [48].
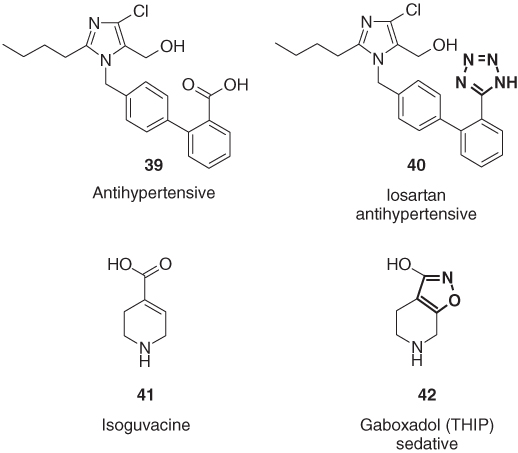
Heterocycles are also capable of mimicking other functional groups, besides the above-mentioned amide group. The most prominent examples are 5-substituted 1H-tetrazole as carboxylic acid replacements [49]. One of the success stories of the tetrazole-carboxylate isosterism is the angiotension II receptor antagonist losartan (40). This drug for the treatment of hypertension and its carboxylic acid lead structure 39 possess similar acidity (pKa of 39: 4.5, losartan: 5.0) but differ significantly in lipophilicity (log P of 39: 1.2, losartan: 4.5). The higher lipophilicity of losartan results in considerably improved oral bioavailability [49]. Also, the two gamma-aminobutyric acid (GABA) agonists isoguvacine (41) and gaboxadol (THIP, 42) possess similar pharmacological properties due to comparable acidity (pKa ≈ 4) (Figure 1.11) [50].
Figure 1.11 The tetrazole derivative losartan (40) and the hydroxyisoxazole derivative gaboxadol (42) as carboxylic acid bioisosteres.
Moreover, triazoles [51], hydroxythiadiazoles [13a], hydroxychromones [52], oxadiazolones [53], and thiazolidinediones [54] have been reported as heterocyclic carboxylic acid bioisosteres.
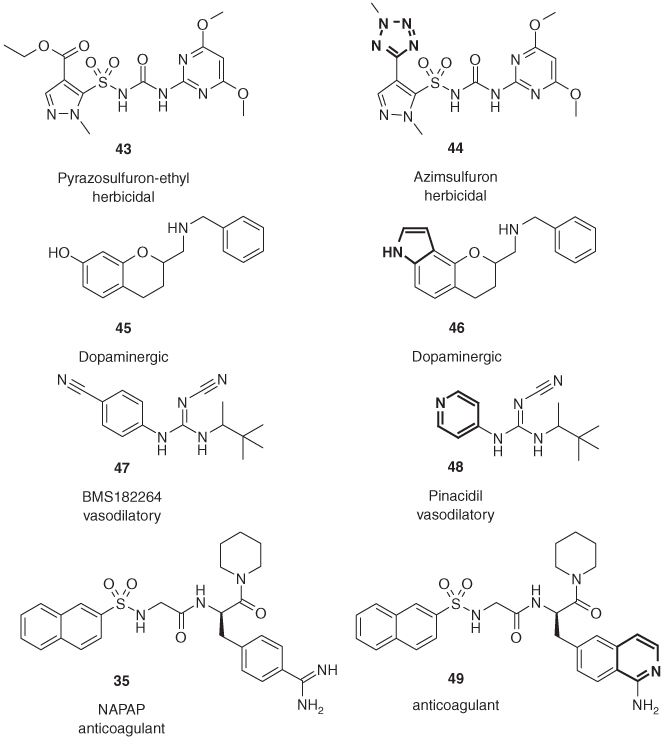
If tetrazole is an excellent carboxylic acid replacement, then alkylated tetrazoles should be able to mimick esters. This is demonstrated by azimsulfuron (44), which shows longer persistence in rice paddy fields than its ethyl ester analog pyrazosulfuron-ethyl (43) [55]. Also, oxazoles [56] and oxadiazoles [57] have been successfully applied as bioisosteres of esters (Figure 1.12).
Figure 1.12 Ring nitrogen atoms of heterocycles 44, 46, 48, and 49 are able to mimic functional groups such as ester, phenol, nitrile, and amidine, respectively.
In search for more potent and selective dopamine D2 agonists for the treatment of psychiatric and neurological diseases such as schizophrenia and Parkinson's disease, the indole moiety in 46 turned out to be an excellent bioisosteric replacement of the metabolically labile phenol function of the lead structure 45 [58].
A widely used trick in lead optimization makes use of the fact that a carbon atom bearing a cyano function is often isosteric with an azomethine, often the ring nitrogen of an aromatic heterocycle. The potassium channel openers BMS182264 (47) and pinacidil (48), only differing by the replacement of a cyanophenyl ring by pyridine are both highly potent aortic smooth muscle relaxants [59].
The replacement of the highly basic benzamidine group in the thrombin inhibitor NAPAP (35) by a moderately basic 1-aminoisoquinoline moiety provides 49, which displays potent enzyme inhibition and significant improvements in membrane transport and oral bioavailability [60].
A phenyl ring in biologically active compounds can often be replaced by a thiophene without any loss of activity because the sulfur atom is equivalent to an ethylenic group with respect to size, mass, and capacity to provide an aromatic lone pair [61]. For instance, a phenyl ring of the biologically active compound piroxicam (50) can be exchanged by thiophene, leading to tenoxicam (51) with similar anti-inflammatory activity (Figure 1.13) [62]. The thiophene derivative sufentanil (53) is at least five times more potent than its phenyl-analog fentanyl (52) [63]. The replacement of the o,o′-dialkylated phenyl ring of the chloroacetamide herbicide metolachlor (54) by a 2,4-dimethylthiophene results in dimethenamid (55) with comparable biological activity [64]. Also, in the area of acetolactate-synthase-inhibiting sulfonylurea herbicides, the ester-substituted phenyl ring could be successfully replaced by thiophene, leading from metsulfuron-methyl (56) to thifensulfuron-methyl (57) [65].
Figure 1.13 The thiophene derivatives 51, 53, 55, and 57 as highly active heterocyclic isosteres of the corresponding phenyl analogs 50, 52, 54, and 56.
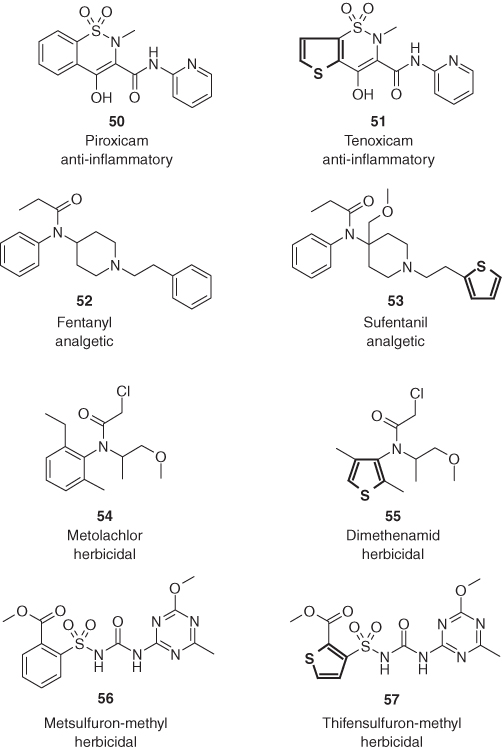
In addition, other heterocycles are able to mimic the phenyl ring of biologically active compounds. The substitution of one of the benzene rings of promazine's phenothiazine scaffold by pyridine led to prothipendyl (59) with improved neuroleptic activity and reduced undesired sedative and extrapyramidal effects (Figure 1.14) [66]. Both compounds are structurally related to the antidepressants maprotiline (60) and imipramine (61), the latter also a heterocyclic isostere of the tetracarbocyclic maprotiline (60) [67]. Interestingly, molecular geometry is determining the direction of pharmacological activity of these four psychotropic drugs [13b]. A dihedral angle between both planes of the two annelated phenyl rings higher than 50°, as is the case for the dibenzobicyclo[2.2.2]octane 60 and the dibenzazepine 61, results in the preponderance of antidepressive activity [68]. If the same angle is only around 25°, as in the phenothiazines 58 and 59, then neuroleptic efficacy prevails.
Figure 1.14 Prothipendyl (59) and imipramine (61), two psychotropic heterocyclic isosteres of promazine (58) and maprotiline (60).
It seems that all kinds of heterocyclic rings, aromatic and nonaromatic ones, can be replaced by other heterocycles, resulting in similar biological activity. The fact that aromatic heterocycles with a similar boiling point are often suitable bioisosteres is an interesting observation [13b]. For instance, a pyridazine (b.p. 208°C) can be replaced successfully by an aromatic heterocycle with one additional ring nitrogen (1,2,4-triazine, b.p. 200°C) but not by a ring with one nitrogen atom less (pyridine, b.p. 115–116°C) or another diazine, in which one of the ring nitrogen is moved to another place (pyrimidine, b.p. 123–124°C, or pyrazine, b.p. 115–118°C) [13b].
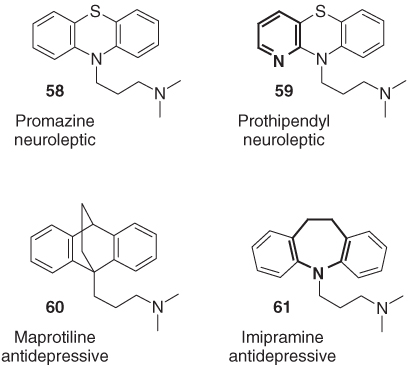
One ring nitrogen and one carbon atom changing places turns a dihydropyrazolo[4,3-d]pyrimidine scaffold into a dihydroimidazo[5,1-f][1,2,4]triazine framework, thereby producing vardenafil (63) from sildenafil (62) [69]. The C-nucleosides oxazofurin (64) and selenazofurin (65) both inhibit the NAD-dependent inosine monophosphate dehydrogenase and show antiviral activities because of the impact of this inhibition of DNA synthesis (Figure 1.15). In contrast to oxazofurin, selenazofurin is also highly active against certain types of leukemia because it is readily metabolized to analogs of NAD, which may be attributed to the higher basicity of selenazole moiety [70]. The muscarinic agonist pilocarpine (66) is widely employed as topical miotic for lowering the elevated intraocular pressure associated with glaucoma, but the duration of this effect lasts only about 3 h, which is mainly due to the hydrolytic instability of the lactone ring. Replacement of one of the chiral carbon atoms in pilocarpine's dihydrofuranone ring by nitrogen results in the cyclic carbamate 67, which is equipotent with pilocarpine and less susceptible to hydrolysis [71]. A ring contraction, which has been successfully applied in pharmaceutical lead optimization, is the replacement of the heptacyclic dihydrobenzodiazepine scaffold of the anticonvulsant α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid (AMPA) receptor antagonist GYKI-53655 (68) by a dihydrophthalazine ring system, as in SYM-2207 (69) [72].
Figure 1.15 The biologically active compounds 63, 65, 67, and 69, bearing a slightly modified heterocycle compared to their analogs 62, 64, 66, and 68.
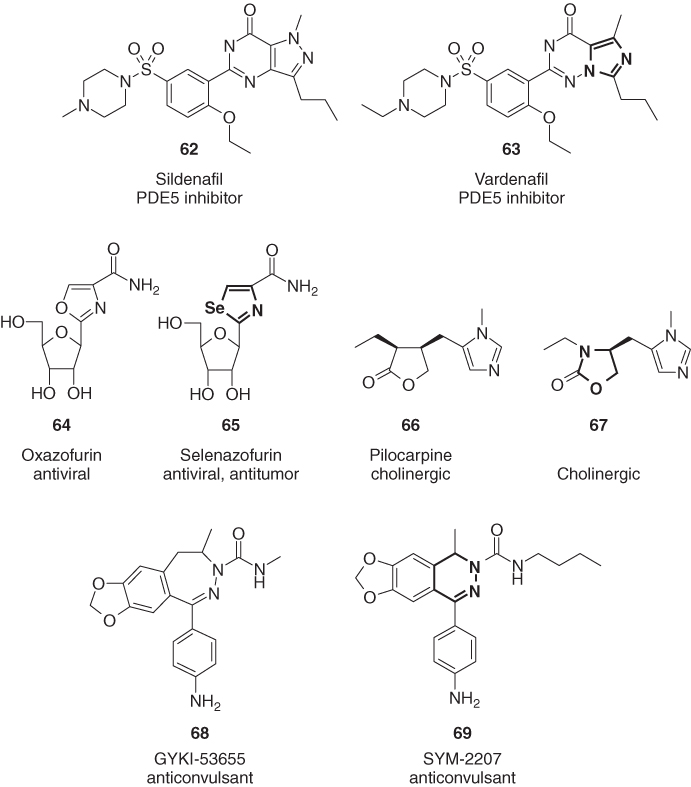
The bleaching herbicide fluridone (70) as well as its tetrahydropyrimidinone analog 71, a cyclic urea, are very active against monocotyledonous and dicotyledonous weeds [73]. In animal health, the triaminated triazine cyromazine (72) and its pyrimidine analog dicyclanil (73) are both very efficient against blowfly strike on sheep and screwworm infestation of cattle [74]. Both compounds are insect growth regulators, inhibiting the biosynthesis of chitin. Both aromatic and aliphatic heterocycles of imidacloprid (74) are replaced in the second-generation neonicotinoid thiamethoxam (75) by an isosteric ring with a different ring size [75]. The sulfonium salt 77, an ionized thiane mimicking successfully the N-protonated fenpropidin (76), which is the active form of this fungicidal sterol biosynthesis inhibitor, shows activity against different wheat phytopathogens (Figure 1.16) [76].
Figure 1.16 The biologically active compounds 71, 73, 75, and 77, bearing a slightly modified heterocycle compared to their analogs 70, 72, 74, and 76.
Notes
* Identically published in both volumes of “Bioactive Heterocyclic Compound Classes”, as different roles of heterocycles in pharmaceuticals and agrochemicals are explained in this introductory chapter.
References
1. Alvarez-Builla, J., Vaquero, J.J., and Barluenga, J. (eds) (2011) Modern Heterocyclic Chemistry, Wiley-VCH Verlag GmbH, Weinheim.
2. Joule, J.A. and Mills, K. (2010) Heterocyclic Chemistry, Wiley-Blackwell, Oxford.
3. Quin, L.D. and Tyrell, J. (2010) Fundamentals of Heterocyclic Chemistry, Wiley-Blackwell, Oxford.
4. Katritzky, A.R., Ramsden, C.A., Joule, J.A., and Zhdankin, V.V. (2010) Handbook of Heterocyclic Chemistry, Elsevier, Amsterdam.
5. Bansal, R.K. (2008) Heterocyclic Chemistry, Anshan, Tunbridge Wells.
6. Eicher, T. and Hauptmann, S. (2003) The Chemistry of Heterocycles, Wiley-VCH Verlag GmbH, Weinheim.
7. Li, J.J. (ed.) (2005) Name Reactions in Heterocyclic Chemistry, John Wiley & Sons, Inc., Hoboken.
8. Li, J.J. and Gribble, G.W. (eds) (2007) Palladium in Heterocyclic Chemistry – A Guide for the Synthetic Chemist, Elsevier, Oxford.
9. Kühl, O. (2010) Functionalised N-Heterocyclic Carbene Complexes, John Wiley & Sons, Ltd, Chichester.
10. Petrov, V.A. (ed.) (2009) Fluorinated Heterocyclic Compounds, John Wiley & Sons, Inc., Hoboken.
11. Roth, B.D. (2002) Prog. Med. Chem., 40, 1–22.
12. Bartlett, D.W., Clough, J.M., Godwin, J.R., Hall, A.A., Hamer, M., and Parr-Dobrzanski, B. (2002) Pest Manag. Sci., 58, 649–662.
13 For reviews on bioisoterism in pharmaceutical and agrochemical lead optimisation see: (a) Meanwell, N.A. (2011) J. Med. Chem., 54, 2529–2591; (b) Ciapetti, P. and Giethlen, B. (2008) in The Practice of Medicinal Chemistry (ed. C.G. Wermuth), Elsevier, Amsterdam, pp. 290–342; (c) Chen, X. and Wang, W. (2003) Ann. Rep. Med. Chem., 38, 333–346; (d) Olesen, P.H. (2001) Curr. Opin. Drug Discov. Dev., 4, 471–478; (e) Patani, G.A. and LaVoie, E.J. (1996) Chem. Rev., 96, 3147–3176; (f) Koyanagi, T. and Haga, T. (1995) in Synthesis and Chemistry of Agrochemicals IV, ACS Symposium Series, Vol. 584 (eds D.R. Baker, J.G. Fenyes, and G.S. Basarab), American Chemical Society, Washington, DC, pp. 15–24.
14. Ward, A., Brogden, R.N., Heel, R.C., Speight, T.M., and Avery, G.S. (1983) Drugs, 26, 468–502.
15. Calderbank, A. and Farrington, J.A. (1984) Drug Chem. Toxicol., 10, 89–106.
16. Brogden, R.N. (1986) Drugs, 32, 60–70.
17. Lamberth, C. (2007) Heterocycles, 71, 1467–1502.
18. Ho, I.K. and Hoskins, B. (1986) Mech. Drug Action, 1, 177–201.
19. Boura, A.L.A. and Green, A.F. (1984) Discov. Pharmacol., 2, 73–105.
20. Jacobsen, N. and Pedersen, L.-E.K. (1983) Pestic. Sci., 14, 90–97.
21. (a) Guile, C.T. (1965) Plant Pathol., 14, 179–187; (b) Munnecke, D.E. and Martin, J.P. (1964) Phytopathology, 54, 941–945.
22. Janssen, P.A.J. (1976) Prog. Drug Res., 20, 347–383.
23. Plosker, G.L. and Benfield, P. (1997) CNS Drugs, 7, 410–416.
24. Kleschick, W.A., Gerwick, B.C., Carson, C.M., Monte, W.T., and Snider, S.W. (1992) J. Agric. Food Chem., 40, 1083–1085.
25. Gates, B.J., Nguyen, T.T., Setter, S.M., and Davies, N.M. (2005) Exp. Opin. Pharmacother., 6, 2117–2140.
26. Kim, D.-S., Chun, S.-J., Jeon, J.-J., Lee, S.-W., and Joe, G.-H. (2004) Pest Manag. Sci., 60, 1007–1012.
27. Haga, T., Fujikawa, K., Sakashita, N., and Nishiyama, R. (1987) Nippon Noyaku Gakkaishi, 12, 311–325.
28. Veber, D.F., Johnson, S.R., Cheng, H.-Y., Smith, B.R., Ward, K.W., and Kopple, K.D. (2002) J. Med. Chem., 45, 2615–2623.
29. Prakash, A. and Jarvis, B. (1999) Drugs, 58, 1137–1164.
30. (a) Pallett, K.E., Cramp, S.M., Little, J.P., Veerasekaran, P., Crudace, A.J., and Slater, A.E. (2001) Pest Manag. Sci., 57, 133–142; (b) Garcia, I., Job, D., and Matringe, M. (2000) Biochemistry, 39, 7501–7507.
31. Lamberth, C. (2004) J. Sulf. Chem., 25, 39–62.
32. (a) Shimizu, T., Hashimoto, N., Nakayama, I., Nakao, T., Mizutani, H., Unai, T., Yamaguchi, M., and Abe, H. (1995) Plant Cell Physiol., 36, 625–632; (b) Sato, Y., Hoshi, T., Iida, T., Ogino, C., Nicolaus, B., Wakabayashi, K., and Boeger, P. (1994) Z. Naturforsch. C, 49, 49–56.
33. For reviews on heterocyclic peptidomimetics see: (a) Abell, A.D. (2002) Lett. Peptide Sci., 8, 267–272; (b) Venkatesan, N. and Kim, B.H. (2002) Curr. Med. Chem., 9, 2243–2270; (c) Bohacek, R.S. and Skakespeare, W.C. (2000) in The Amide Linkage: Selected Structural Aspects in Chemistry, Biochemistry and Material Science (eds A. Greenberg, C.M. Breneman, and J.F. Liebman), John Wiley & Sons, Ltd, Chichester, pp. 377–407; (d) Giannis, A. and Kolter, T. (1993) Angew. Chem., 105, 1303–1326; (1993) Angew. Chem., Int. Ed. Engl., 32, 1244–1267.
34. La Porte, C.J.L. (2009) Exp. Opin. Drug Metabol. Toxicol., 5, 1313–1322.
35. May, B.C.H. and Abell, A.D. (2002) J. Chem. Soc., Perkin Trans. 1, 172–178.
36. Getman, D.P., DeCrescenzo, G.A., Heintz, R.M., Reed, K.L., Talley, J.J., Bryant, M.L., Clare, M., Houseman, K.A., Marr, J.J., Mueller, R.A., Vazquez, M.L., Shieh, H.-S., Stallings, W.C., and Stegeman, R.A. (1993) J. Med. Chem., 36, 288–291.
37. (a) Thompson, S.K., Murthy, K.H.M., Zhao, B., Winborne, E., Green, D.W., Fisher, S.M., DesJarlais, R.L., Tomaszek, T.A., Meek, T.D., Gleason, J.G., and Abdel-Meguid, S.S. (1994) J. Med. Chem., 37, 3100–3107; (b) Thompson, S.K., Eppley, A.M., Frazee, J.S., Darcy, M.G., Lum, R.T., Tomaszek, T.A., Ivanoff, L.A., Morris, J.F., Sternberg, E.J., Lambert, D.M., Fernandez, A.V., Petteway, S.R., Meek, T.D., Metcalf, B.W., and Gleason, J.G. (1994) Bioorg. Med. Chem. Lett., 4, 2441–2446.
38. (a) Smith, A.B., Hirschmann, R., Pasternak, A., Yao, W., Sprengeler, P.A., Holloway, M.K., Kuo, L.C., Chen, Z., Darke, P.L., and Schleif, W.A. (1997) J. Med. Chem., 40, 2440–2444; (b) Gosh, A.K., Thompson, W.J., McKee, S.P., Duong, T.T., Lyle, T.A., Chen, J.C., Darke, P.L., Zugay, J.A., Emini, E.A., Schleif, W.A., Huff, J.R., and Anderson, P.S. (1993) J. Med. Chem., 36, 292–294.
39. Mack, H., Pfeiffer, T., Hornberger, W., Bohm, H.-J., and Hoeffken, H.W. (1995) J. Enzyme Inhib., 9, 73–86.
40. (a) Saitton, S., Del Tredici, A.L., Saxin, M., Stenström, T., Kihlberg, J., and Luthman, K. (2008) Org. Biomol. Chem., 6, 1647–1654; (b) Saitton, S., Del Tredici, A.L., Mohell, N., Vollinga, R.C., Boström, D., Kihlberg, J., and Luthman, K. (2004) J. Med. Chem., 47, 6595–6602.
41. (a) Bolton, D., Boyfield, I., Coldwell, M.C., Hadley, M.S., Johns, A., Johnson, C.N., Markwell, R.E., Nash, D.J., Riley, G.J., Scott, E.E., Smith, S.A., Stemp, G., Wadsworth, H.J., and Watts, E.A. (1997) Bioorg. Med. Chem. Lett., 7, 485–488; (b) Van Wijngaarden, I., Kruse, C.G., van Hes, R., van der Heyden, J.A.M., and Tulp, M.T.M. (1987) J. Med. Chem., 30, 2099–2104.
42. (a) Steinmetzer, T., Zhu, B.Y., and Konishi, Y. (1999) J. Med. Chem., 42, 3109–3115; (b) Mimoto, T., Kato, R., Takaku, H., Nojima, S., Terashima, K., Misawa, S., Fukazawa, T., Ueno, T., Sato, H., Shintani, M., Kiso, Y., and Hayashi, H. (1999) J. Med. Chem., 42, 1789–1802.
43. Kim, B.H., Chung, Y.J., Keum, G., Kim, J., and Kim, K. (1992) Tetrahedron Lett., 33, 6811–6814.
44. Jones, R.C.F. and Ward, G.J. (1988) Tetrahedron Lett., 29, 3853–3856.
45. Einsiedel, J., Thomas, C., Hübner, H., and Gmeiner, P. (2000) Bioorg. Med. Chem. Lett., 10, 2041–2044.
46. Liu, Y., Li, Y., Myles, D.C., Claypool, M., Carreras, C.W., and Shaw, S.J. (2010) Bioorg. Med. Chem., 18, 7651–7658.
47. (a) Legeay, J.C., Vanden Eynde, J.J., and Bazureau, J.P. (2007) Tetrahedron Lett., 48, 1063–1068; (b) Feng, D.D., Biftu, T., Candelore, M.R., Cascieri, M.A., Colwell, L.F., Deng, L., Feeney, W.P., Forrest, M.J., Hom, G.J., MacIntyre, D.E., Miller, R.R., Stearns, R.A., Strader, C.D., Tota, L., Wyvratt, M.J., Fisher, M.H., and Weber, A.E. (2000) Bioorg. Med. Chem. Lett., 10, 1427–1429; (c) Andersen, K.E., Jorgensen, A.S., and Braestrup, C. (1994) Eur. J. Med. Chem., 29, 393–399.
48. (a) Zhang, D., Wang, Z., Xu, W., Sun, F., Tang, L., and Wang, J. (2009) Eur. J. Med. Chem., 44, 2202–2210; (b) Chen, J.J., Zhang, Y., Hammond, S., Dewdney, N., Ho, T., Lin, X., Browner, M.F., and Castelhano, A.L. (1996) Bioorg. Med. Chem. Lett., 6, 1601–1606.
49. For reviews on tetrazoles as carboxylic acid bioisosteres see: (a) Zych, A.J. and Herr, R.J. (2007) PharmaChem, 6, 21–24; (b) Herr, R.J. (2002) Bioorg. Med. Chem., 10, 3379–3393.
50. (a) Frolund, B., Ebert, B., Kristiansen, U., Liljefors, T., and Krogsgaard-Larsen, P. (2002) Curr. Top. Med. Chem., 2, 817–832; (b) Krogsgaard-Larsen, P., Hjeds, H., Falch, E., Jorgensen, F.S., and Nielsen, L. (1988) in Advances in Drug Research (ed. B. Testa), Academic Press, London, pp. 381–456.
51. Drysdale, M.J., Pritchard, M.C., and Horwell, D.C. (1992) J. Med. Chem., 35, 2573–2581.
52. Atkinson, J.G., Girard, Y., Rokach, J., Rooney, C.S., McFarlane, C.S., Rackham, A., and Share, N.N. (1979) J. Med. Chem., 22, 99–106.
53. (a) Gezginci, M.H., Martin, A.R., and Franzblau, S.G. (2001) J. Med. Chem., 44, 1560–1563; (b) Kohara, Y., Kubo, K., Imamiya, E., Wada, T., Inada, Y., and Naka, T. (1996) J. Med. Chem., 39, 5228–5235; (c) Bock, M.G., DiPardo, R.M., Mellin, E.C., Newton, R.C., Veber, D.F., Freedman, S.B., Smith, A.J., Patel, S., Kemp, J.A., Marshall, G.R., Fletcher, A.E., Chapman, K.L., Anderson, P.S., and Freidinger, R.M. (1994) J. Med. Chem., 37, 722–724.
54. (a) Henke, B.R. (2004) J. Med. Chem., 47, 4118–4127; (b) Hulin, B., McCarthy, P.A., and Gibbs, E.M. (1996) Curr. Pharm. Des., 2, 85–102.
55. Marquez, T., Joshi, M.M., Fader, T.P., and Massasso, W. (1995) Brighton Crop Prot. Conf. Weeds, 1, 65–72.
56. Kim, K.S., Kimball, S.D., Misra, R.N., Rawlins, D.B., Hunt, J.T., Xiao, H.-Y., Lu, S., Qian, L., Han, W.-C., Shan, W., Mitt, T., Cai, Z.-W., Poss, M.A., Zhu, H., Sack, J.S., Tokarski, J.S., Chang, C.Y., Pavletich, N., Kamath, A., Humphreys, W.G., Marathe, P., Bursuker, I., Kellar, K.A., Roongta, U., Batorsky, R., Mulheron, J.G., Bol, D., Fairchild, C.R., Lee, F.Y., and Webster, K.R. (2002) J. Med. Chem., 45, 3905–3927.
57. (a) Petukhov, P.A., Zhang, M., Johnson, K.J., Tella, S.R., and Kozikowski, A.P. (2001) Bioorg. Med. Chem. Lett., 11, 2079–2083; (b) Sauerberg, P., Kindtler, J.W., Nielsen, L., Sheardown, M.J., and Honore, T. (1991) J. Med. Chem., 34, 687–692; (c) Orlek, B.S., Blaney, F.E., Brown, F., Clark, M.S.G., Hadley, M.S., Hatcher, J., Riley, G.J., Rosenberg, H.E., Wadsworth, H.J., and Wyman, P. (1991) J. Med. Chem., 34, 2726–2735; (d) Street, L.J., Baker, R., Book, T., Kneen, C.O., MacLeod, A.M., Merchant, K.J., Showell, G.A., Saunders, J., Herbert, R.H., Freedman, S.B., and Harley, E.A. (1990) J. Med. Chem., 33, 2690–2697; (e) Saunders, J., Cassidy, M., Freedman, S.B., Harley, E.A., Iversen, L.L., Kneen, C., MacLeod, A.M., Merchant, K.J., Snow, R.J., and Baker, R. (1990) J. Med. Chem., 33, 1128–1138.
58. Mewshaw, R.E., Zhao, R., Shi, X., Marquis, K., Brennan, J.A., Mazandarani, H., Coupet, J., and Andree, T.H. (2002) Bioorg. Med. Chem. Lett., 12, 271–274.
59. (a) Butera, J.A., Antane, M.M., Antane, S.A., Argentieri, T.M., Freeden, C., Graceffa, R.F., Hirth, B.H., Jenkins, D., Lennox, J.R., Matelan, E., Norton, N.W., Quagliato, D., Sheldon, J.H., Spinelli, W., Warga, D., Wojdan, A., and Woods, M. (2000) J. Med. Chem., 43, 1187–1202; (b) Atwal, K.S., Moreland, S., McCullough, J.R., O'Reilly, B.C., Ahmed, S.Z., and Normandin, D.E. (1992) Bioorg. Med. Chem. Lett., 2, 83–86.
60. Rewinkel, J.B.M., Lucas, H., van Galen, P.J.M., Noach, A.B.J., van Dinther, T.G., Rood, A.M.M., Jenneboer, A.J.S.M., and van Boeckel, C.A.A. (1999) Bioorg. Med. Chem. Lett., 9, 685–690.
61. (a) Press, J.B. (1991) Chem. Heterocycl. Comp., 44, 397–502; (b) Drehsen, G. and Engel, J. (1983) Sulfur Rep., 3, 171–214.
62. Bird, H.A. and Naden, M.A. (1989) J. Drug Dev., 2, 119–135.
63. Monk, J.P., Beresford, R., and Ward, A. (1988) Drugs, 36, 286–313.
64. Couderchet, M., Bocion, P.F., Chollet, R., Seckinger, K., and Böger, P. (1997) Pestic. Sci., 50, 221–227.
65. Cuomo, J., Gee, S.K., and Hartzell, S.L. (1991) in Synthesis and Chemistry of Agrochemicals II, ACS Symposium Series, Vol. 443 (eds D.R. Baker, J.G. Fenyes, and W.B. Moberg), American Chemical Society, Washington, DC, pp. 62–73.
66. Ellenbroek, B., Prinssen, E., and Cools, A. (1992) Neurosci. Res. Commun., 11, 155–161.
67. Gurguis, G.N., Blakeley, J.E., Antai-Otong, D., Vo, S.P., Orsulak, P.J., Petty, F., and Rush, A.J. (1999) J. Psychiatr Res., 33, 309–322.
68. Wilhelm, M. (1975) Pharm. J., 214, 414–416.
69. Stamford, A.W. (2002) Ann. Rep. Med. Chem., 37, 53–64.
70. (a) Franchetti, P., Cristalli, G., Grifantini, M., Cappellacci, L., Vittori, S., and Nocentini, G. (1990) J. Med. Chem., 33, 2849–2852; (b) Goldstein, B.M., Takusagawa, F., Berman, H.M., Srivastava, P.C., and Robins, R.K. (1985) J. Am. Chem. Soc., 107, 1394–1400.
71. (a) Hobbs, S.H., Johnson, S.J., Kesten, S.R., Pavia, M.R., Davis, R.E., Schwarz, R.D., Coughenour, L.L., Myers, S.L., Dudley, D.T., and Moos, W.H. (1991) Bioorg. Med. Chem. Lett., 1, 147–150; (b) Sauerberg, P., Chen, J., WoldeMussie, E., and Rapoport, H. (1989) J. Med. Chem., 32, 1322–1326.
72. Pelletier, J.C., Hesson, D.P., Jones, K.A., and Costa, A.-M. (1996) J. Med. Chem., 39, 343–346.
73. (a) Babczinski, P., Blunck, M., Sandmann, G., Shiokawa, K., and Yasui, K. (1995) Pestic. Biochem. Physiol., 52, 45–59; (b) Babczinski, P., Sandmann, G., Schmidt, R.R., Shiokawa, K., and Yasui, K. (1995) Pestic. Biochem. Physiol., 52, 33–44; (c) Babczinski, P., Blunck, M., Sandmann, G., Schmidt, R.R., Shiokawa, K., and Yasui, K. (1990) Pestic. Sci., 30, 339–342.
74. (a) Anziani, O.S., Guglielmone, A.A., and Schmid, H. (1998) Vet. Parasitol., 76, 229–232; (b) Hart, R.J., Cavey, W.A., Ryan, K.J., Strong, M.B., Moore, B., Thomas, P.L., Boray, J.C., and von Orelli, M. (1982) Aust. Vet. J., 59, 104–109.
75. (a) Maienfisch, P., Angst, M., Brandl, F., Fischer, W., Hofer, D., Kayser, H., Kobel, W., Rindlisbacher, A., Senn, R., Steinemann, A., and Widmer, H. (2001) Pest Manag. Sci., 57, 906–913; (b) Maienfisch, P., Huerlimann, H., Rindlisbacher, A., Gsell, L., Dettwiler, H., Haettenschwiler, J., Sieger, E., and Walti, M. (2001) Pest Manag. Sci., 57, 165–176.
76. Wilkie, J.S. and Winzenberg, K.N. (1992) Aust. J. Chem., 45, 457–461.
Part I
Herbicides
2
Triazine Herbicides
This chapter deals with triazine products and their use in agriculture. As the name suggests, triazines are aromatic heterocycles containing three nitrogen atoms, and as such, there are three triazine regioisomers possible (Figure 2.1).
Figure 2.1 Triazine regioisomers.
Although 1,2,4- and 1,2,3-triazines can be found in a few agrochemical products [1, 2], the 1,3,5-triazines (often called sym-triazines or s-triazines) constitute by far the most important class.
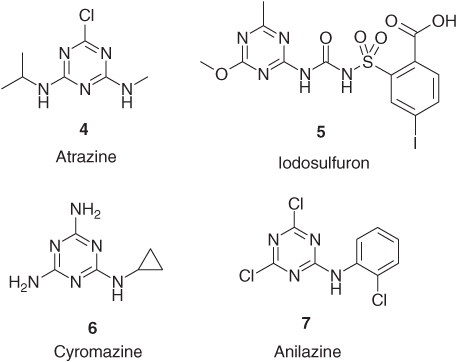
The s-triazines find broad application in herbicides, for example, photosystem II (PS II) inhibitors [3] such as atrazine (4, Gesaprim®, Syngenta) and sulfonylurea acetolactate synthase inhibitors [4] such as iodosulfuron (5, Husar®, Bayer CropScience) and to a much lesser extent, insecticides [5] such as cyromazine (6, Trigard®, Syngenta) and fungicides [6] such as anilazine (7, Dyrene®, Bayer CropScience, Figure 2.2) [6]. This short review particularly concentrates on the atrazine-type herbicides [7].
Figure 2.2 Examples of 1,3,5-triazine containing agrochemicals.
Much of the triazine chemistry developed for application in agrochemistry (and other fields for that matter!) is based on the chemistry of cyanuric chloride. Cyanuric chloride (8) is a commodity chemical produced by the catalytic trimerization of cyanogen chloride, the latter being produced by the reaction of chlorine and hydrocyanic acid (Scheme 2.1) [2]. Cyanuric chloride, with its three chlorines being replaceable in a stepwise SNAr manner by, for example, carbon, nitrogen, oxygen, or sulfur nucleophiles, is a chemists dream for production of a vast variety of molecules.
Scheme 2.1 Synthesis of cyanuric chloride and its reactivity with nucleophiles.

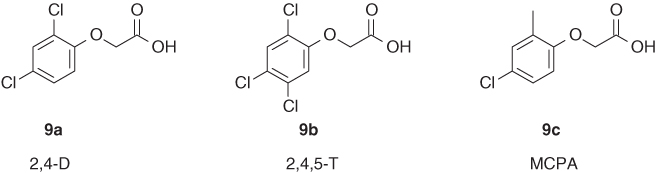
An empirical rule for amine substituents states that the first chlorine is replaced at 0–5°C, the second at 30–50°C, and the third at 70–100°C [8]. Reactions are typically carried out in the presence of HCl scavengers that may be organic or inorganic. Not surprisingly, many of the products from such reactions have found industrial applications in many fields too long to list here but well documented in Ref. [2]. So how did the triazines become so important in the field of PS II inhibitors? In the early 1950s, the scientists at J. R. Geigy (a legacy company of Syngenta) had decided to evaluate farmers' needs for weed control [9]. At that time, the market was dominated by the phenoxyacetic acid herbicides such as 2,4-D (9a), 2,4,5-T (9b), and 2-methyl-4-chlorophenoxyacetic acid (MCPA) (9c, Figure 2.3).
Figure 2.3 Phenoxyacetic acid herbicides.
Scheme 2.210