

The Science and Art
of Dental Ceramics
Volume I: The Nature of Dental Ceramics
and their Clinical Use
Consulting Professor
in Fixed Prosthodontics and Biomaterials
Louisiana State University Medical Centre School
of Dentistry

Quintessence Publishing Co., Inc. 1979
Chicago, Berlin, Rio de Janeiro, Tokyo

© 1979 by Quintessence Publishing Co., Inc., Chicago, Illinois.
All rights reserved.
This book or any part thereof must not be reproduced by any means or in any form without the written permission of the publisher.
Lithography: Industrie- und Presseklischee, Berlin.
Composition: Westkreuz-Druckerei Berlin/Bonn.
Printing and binding: North Central Publishing Co., St. Paul.
Printed in the U.S.A.
ISBN 0 931386 04 7
Foreword
Dr. John McLean’s practice of aesthetic dentistry goes back many years. In 1963 he initiated a programme of research with the Warren Spring Government Laboratory in England with the object of developing stronger dental porcelains without sacrificing aesthetics. This programme led to the development of the Aluminous Porcelains which have largely supplanted regular dental porcelain for the construction of jacket crowns.
Dr. McLean is one of a handful of individuals endowed with exceptional clinical talent and the scientific ability to produce an aesthetic restoration from the basic raw ingredients.
This series of monographs is a new endeavour to present, in detail, the finer points of ceramic art in dentistry. It is our intention at Louisiana State University School of Dentistry to use this work as the basis for a continuing education programme in dental ceramics which we hope will encourage younger men to specialise in this rapidly expanding field.
Edmund Jeansonne
Dean
November 1974
Preface
This series of monographs on “The Science and Art of Dental Ceramics” has been written principally for the serious student or researcher in dental ceramics. They may also assist the dental technician in a better understanding of his craft and will explain why failures may occur during the firing of dental ceramic work.
Each monograph attempts to penetrate, in some depth, the present state of the art, and some of the material is not available in current textbooks or research papers in the dental field. The research student might find the gathering together of the newer research in dental porcelain of some assistance if he is preparing a thesis. It is for this reason that the Monographs were originally published by Louisiana State University as two separate books.
The increasing demand for information on dental ceramics has continued since the publication of the Monographs in 1974 and the Quintessence Publishing Company considered it an opportune moment to revise the text and publish it in one volume.
The scientific section has been updated particularly with regard to the latest developments in the metal-ceramic field. Two major symposia have been held in the United States of America in 1977 on dental porcelain and alternatives to gold alloys. The proceedings were published by the University of Southern California and the National Institute of Health.
Reference is made to these symposia in the text and the clinical section of the book has been enhanced by the inclusion of colour photographs which do so much for the beauty of porcelain work.
The first two monographs are concerned with the nature of dental ceramics and glasses and how these brittle materials may be strengthened or prevented from fracturing. Further monographs deal with the problems of colour, occlusion in dental porcelain, and the preparation of teeth for ceramic crown and bridgework. The building of ceramics with a brush and methods of obtaining colour and translucency are also dealt with in detail. The importance of obtaining this natural enamel effect cannot be over-emphasized since much of the ceramic work today lacks the natural depth of translucency of human teeth. Without depth of translucency, ceramic crowns will never become submerged in the mouth and be undetectable from their human counterparts.
The increasing use of metal-ceramics has brought with it the problems of maintaining the health of the periodontium. In addition, the problems of dealing with highly reflective opaque backgrounds on metal surfaces is one that has yet to be completely mastered. The maintenance of original tooth contour, together with the provision of correct colour values, is an area which is given great prominence in this series of monographs; this work forming part of the continuing education programme at Louisiana State University.
No attempt has been made in this volume to cover all aspects of fixed prosthodontics and the reader is advised to consult other works in this field. For a basic understanding of crown and bridgework the text by Shillingburg, Hobo and Whitsett “Fundamentals of Fixed Prosthodontics” is recommended. Other works that provide additional instruction are Johnston, Phillips and Dykema’s “Modern Practice in Crown and Bridge Prosthodontics” and Tylman’s “Theory and Practice of Crown and Bridge Prosthodontics”.
The design and construction of metal ceramic bridgework and developing occlusion in dental porcelain will be covered in the second volume of “The Science and Art of Dental Ceramics” entitled “Laboratory Procedures in Dental Ceramics”. In this volume a critical look is taken of obtaining maximum light tranmission through the anterior crown. Many students are being instructed only in the metal-ceramic techniques and therefore tend to accept the aesthetics of these materials without question. This has resulted in many commercial laboratories eliminating the porcelain jacket crown from their armamentarium. The “metal-ceramic smile” is more common than we perhaps would care to admit.
The cast metal-ceramic restoration is of enormous value in dentistry and is deservedly the most widely used porcelain restoration. However, a plea is made in these monographs for more selectivity in the use of porcelain materials. To adopt one system or brand of porcelain and use it indiscriminately and without appreciation of its aesthetic or mechanical limitations can result in unsightly dentistry. There is a place for all our materials – the full porcelain veneer crown, the bonded alumina crown, and the metal ceramic crown. Each should be used where its properties can be developed to maximum advantage.
It should be noted that throughout the text, the new Système International d’Unités (SI) is used. The SI derives all the quantities needed in all technologies from only six basic and arbitrarily defined units. This contrasts with the metric systems currently used, in which additional quantities (for instance, ‘calorie’ and ‘horsepower’) are arbitrarily and, indeed, differently defined in different metric countries. Relationships between units are thus greatly simplified in the SI, the introduction of which offers existing metric countries a unique opportunity to harmonize their measuring practices. It is likely that most countries will eventually adopt this international system of measurement and, for convenience, a conversion table is inserted at the end of each volume.
The Harvard System of references has been chosen quite deliberately since it requires the insertion in the text of the name of the author quoted as a reference. The reader is then able to identify the person quickly and realise who had developed the original thought.
Acknowledgements
In preparing this series of monographs on the “Science and Art of Dental Ceramics” I have had considerable help from experts in the fields of ceramics, metallurgy, and silicate chemistry.
In particular I would like to thank Mr. David Binns of the British Ceramic Research Association, Dr Alan Wilson of the Laboratory of the Government Chemist, Mr. R. Batchelor of Harrison Meyer Ltd., and Dr. Ian Sced of the National Physical Laboratory, for assisting me with the sections on the nature of glasses, ceramics and metals.
I have had the good fortune of working with Mr. T. H. Hughes of the Warren Spring Laboratory, Stevenage, on the development of the aluminous porcelains. Without his help these monographs would not have been possible.
A great deal of my knowledge on porcelain has been gained during the production of the aluminous porcelains and I am greatly in debt to Mr. Clifford Hall of Harrison Meyer Ltd., Stokeupon-Trent, who has always allowed me free access to his laboratories and technical staff. I am also indebted to Dr. H. Rauter, K. G., of Vita Zahnfabrik, West Germany, for his co-operation in producing alumina reinforced porcelains, and to Dr. Pralow, his chief chemist, with whom I have worked for a number of years.
The late Mr. Jan Adriaansen of Amsterdam taught me how to build porcelain and he has given considerable inspiration to my technicians.
Mr. Richard Gale, Mr. John Seviour, Mr. Michael Kempton, Mr. John Hubbard and Mr. Michael Kedge have performed much of the work on the aluminous porcelains and their willing assistance materially improved the techniques of construction.
Mrs. K. Wilder has given invaluable service during the preparation of my manuscripts and my daughter, Diana, has prepared all the line drawings. Mr. James Morgan and his staff of the photographic department at the Institute of Dental Surgery, Eastman Dental Hospital, have prepared most of the other illustrations and I am greatly indebted to all of them for their years of painstaking work.
The production of this work has been possible only with the help and encouragement of Dean Jeansonne and his staff at Louisiana State University Dental School. In particular, the advice of Dr. Howard Bruggers on the format of the monographs and his editorial help is greatly appreciated. I would also like to thank Mr. Raymond Calvert, Mrs. Mary U. Fontenot, Mrs. Linda S. Garner, Mr. William Stallworth, Mr. Barry Morgan, Mr. Alex Barkoff, Mrs. Christine Taylor, Mrs. Areme Kavanaugh and Miss Michelle Jilek, of the Learning Resources Department who have been responsible for the production of these monographs.
John W. McLean
38 Devonshire street
London, WIN 1LD
Contents
Cover
Table of Contents
Foreword
Preface
Acknowledgements
Introduction
Monograph I – The Nature of Dental Ceramics
Crystalline Ceramics
Extraction of Alumina
Fabrication of Alumina Components
Sintering of Alumina
Translucent Alumina
Effect of Debasing Alumina
Colouring Alumina
Dental Alumina
Physical Properties
The Nature of Glasses
Glass Formation
Formulation of Dental Porcelain
The Role of Fluxes
Intermediate Oxides
Boric Oxide Fluxes
Chemical Compositon of Dental Porcelain
Devitrification and Thermal Expansion
Fritting
Alternative Additives to Dental Porcelain
Felspathic Porcelain
Nepheline Syenite
Colouring and Opacifying Dental Porcelain
Colour Pigments
Opacifying Agents
Stains and Colour Modifiers
Fluorescence
Glazes and Add-on Porcelain
Classification of Dental Porcelains
Porcelains Used in Tooth Manufacture
Condensation of Dental Porcelain
Volume Porosity of Powders
Condensation
Surface Tension
The Wet Brush Technique
Sintering of Dental Porcelains
Air Firing Porcelain
Particle Size and Translucency
Vacuum-firing-Porcelains
Diffusible Gas Firing Process
Pressure Cooling Process
Classification of Stages of Maturity
Glazing and Thermal Shock Prevention
Thermal Shock
Solubility
References
Monograph II – The Strengthening of Dental Porcelain
Crack Propagation in Dental Porcelain
Mechanical Testing
Testing Ground Porcelain Specimens
Testing Glazed Specimens
Strength of Glazed and Surface Ground Porcelain
Influence of Condensation Techniques on Mechanical Strength
Methods of Strengthening Dental Porcelain
Enamelling of Metals
Types of Metal-Ceramic Systems
Noble-Metal Alloy Systems
Base-Metal Alloy Systems
Gold-Platinum-Palladium Alloys
Gold-Platinum-Palladium Alloys-Mechanical Strength and Heat Treatment
Gold Bonding Agents
Alternative Alloy Systems
Gold-Platinum-Tungsten Alloys
Gold-Palladium-Silver Alloys
Palladium-Silver Alloys
Mechanical properties of the noble-metal alloys
Nature of the Porcelains used in Metal-Ceramic Systems
The Nature of the Metal-Ceramic Bond
Methods of Testing Bond Strength
Choice of Test Piece Design
Results of Testing Bond Strengths
Technical Considerations
Noble-Metal Alloy Systems
Base Metal Alloy/Porcelain Systems
Bond Strength of Base Metal Alloy/Porcelain Systems
Types of Metal/Porcelain Failure
1. Metal-Porcelain
2. Metal Oxide-Porcelain
3. Metal-Metal Oxide
4. Metal Oxide-Metal Oxide
5. Cohesive within Metal
6. Cohesive within Porcelain
Effect of the Degree of Oxidation and Cooling Rate on the Strength of the Bond
Technical Considerations-Base-Metal Alloy Systems
Evaluation of Metal-Ceramic Systems
Oxide Formation
Strength and Castability
1. Yield Strength
2. Modulus of Elasticity
3. Creep or “sag-resistance”
4. Hardness
5. Burnishability
Gold-platinum-palladium alloys
Gold-palladium-silver alloys
Palladium-silver alloys
Base-metals Nickel-chromium alloys
Bonding of Porcelain to Precious Metals Using Tin Oxide Coatings
The Platinum Bonded Alumina Crown
Cementation
Dispersion Strengthening of Glasses
Types of Reinforcing Crystals
Types of Alumina
Calcined Alumina
Fused Alumina
Types of Glass used with Alumina
Aluminous Porcelain
Aluminous Porcelain Core Powders
Influence of Grain Size on Opacity and Strength
Influence of Alumina Crystal Concentration on Strength
Crack-propagation in Aluminous Porcelain
Sintering Aluminous Porcelain
Effect of Sintering Time on the Strength of Aluminous Porcelain
Dispersion Strengthening of Glass with Alumina Whiskers
Enamelling of High Strength Crystalline Ceramics
Strength of Alumina-Enamel Laminates
Bonding of Aluminous Porcelain to High Alumina
Controlled Crystallisation of Glasses
Production of Pre-stressed Surface Layers in Dental Porcelain via Ion-exchange
Technical Considerations
Aluminous Porcelains
References
Monograph III – Aesthetics of Dental Porcelain
The Nature of Light
Primary Colours
The Three Dimensions of Colour
Hue
Chroma
Value
Colour Measurement
Complimentary Colours
Optical Properties
Reflection
Regular or Specular Reflection
Diffuse Reflection
Refractive index and dispersion
Reflection and Refraction
Light Scattering
Opacity
Paint-on-Opaques
Translucency
Surface Gloss
Effect of Metal Surface Treatment on the Masking Power of Opaque Porcelain
Ultra-Violet Radiation and Fluorescence
Radio-active compounds in dental porcelain
Radiation hazards
Colour Blending
Colour Production in Natural Teeth
Incisal Third
Middle Third
Cervical Third
Reproducing Natural Teeth in Dental Porcelain
Types of Porcelain Crowns
Felspathic crown – air-fired
Felspathic crown – vacuum-fired
The Metal-Ceramic Crown
Metamerism
The Aluminous Porcelain Crown
The Role of Opaque Porcelains in Obtaining Aesthetics
Requirements for a Tooth Shade Guide
The Munsell Colour Order System
The Adams Co-ordinate System
Colour of Natural Teeth related to Modern Shade Guides
Shade Matching
The Custom-Built Shade Guide
The Custom-Built Metal-Ceramic Shade Guide
Vacuum-Fired Porcelain Tooth Shade Guides
The Eye and Colour Perception
Recommended Procedures for Shade Matching
Determing Hue
Determining Chroma
Determining Value
Determining Metameric Effects
Blending of Colours
Custom staining
The Study Cast
The Influence of Tooth Contour on Aesthetics
Vertical correction
Horizontal correction
Instanding and Outstanding Teeth
Rotated Teeth
Significance of intra-arch space
Spaced teeth
Mesial or distal inclination of teeth
Outline form and size
Making a tooth look wider or narrower
Making a tooth look shorter or longer
Orthodontic Correction
Reproducing Tooth Anatomy in Dental Porcelain
Common faults in porcelain tooth anatomy
The Central incisors
Lateral Incisors
The Canine Teeth
The Posterior Teeth
Full Mouth Reconstruction
The repeated ratio
Front-to-back progression
Horizontal and Vertical space allocation
References
Selected Reading
Monograph IV – Porcelain as a Restorative Material
Plaque Accumulation on Restorative Materials
Marginal Fit
Micro-leakage under Porcelain Crowns
Occlusion and Dental Porcelain
Gold versus Porcelain Occlusion
Indications for use
Indications for Use of Porcelain Jacket Crowns
Contra-lndications for Use of the Porcelain Jacket Crown
Indications for use of the Metal-Ceramic Crown
Contra-Indications for use of the Metal-Ceramic Crown
Aluminous Porcelain Compared with Metal-Ceramics
Aluminous Porcelain
Metal-Ceramics
The Platinum bonded Alumina Crown
Indications for use of the Bonded Alumina Crown
Contra-indications
Clinical Cases
The Complete Porcelain Veneer Crown and the Platinum Bonded Alumina Crown
Aesthetics
Strength
Periodontal Health and Occlusion
Health of the Pulp
Preserving Pulp Vitality
The Devitalised Tooth
The Preparation
The Typical Preparation
Design Factors
Application to the Metal-Ceramic Crown
The Porcelain Veneer Crown
Stress Analysis and Design Factors
Cervical area and shoulder requirements – Maxillary central incisor
Design Factor – Cervical and Shoulder area – Maxillary central incisor
The lingual and labial surfaces and length of preparation – Maxillary central incisor
Design factor – Lingual and labial surfaces – Maxillary central incisor
Approximal axial walls – Maxillary central incisor
Application to the Metal-Ceramic Crown
Design factor – Approximal Axial Walls – Maxillary central incisor
Shoulder geometry
Definitions
Design factor – Shoulder geometry
Retention
Design factor – Retention
Cement film thickness
The Bonded Alumina Crown and the Complete Porcelain Veneer Crown or Aluminous Porcelain Crown
Recommended dimensionsfor tooth preparation
Stress Analysis of the Posterior Molar Crown
The Premolar Porcelain Veneer Crown
The Molar Porcelain Veneer Crown
The Atypical Preparation
Class 1: Crowns larger than the anatomical average
Class 2: Crowns smaller than anatomical average
Class 3: Crowns which show marked anatomical deviations from normal
Class 4: Crowns with loss of enamel and dentine on either the mesial or distal surfaces
Class 5: Crowns with loss of enamel and dentine on both the mesial and distal surfaces
Class 6: Crown with loss of enamel and dentine at the incisal edge
Class 7: Crowns with loss of enamel and dentine at the cervical margins
Class 8: Crowns with generalised loss of surface enamel
Class 9: Length of clinical crown greater than the anatomical crown
Crown-Root ratio
Summary of the requirements of the Atypical Preparation
The Typical Maxillary Central Incisor Preparation
Registration of surface characterisations of tooth
Shade Determination
Elimination of gingival trauma
Depth of Shoulder
The Preparation
Theoretical Considerations
Cervical approach
Measurements required for establishing the design of preparation
The stages in preparation
Operative technique
Instrumentation
Vario-air coupling (Kavo Super-torque)
Labial and Lingual Depth Cuts
Reduction of the Lingual Surface
Reduction of the Labial Surface
Relationship of the shoulder to the interdental papilla
Creation of the Labial Sub-Gingival Shoulder
Reduction of Incisal Edge
Monitoring the Depth of the Preparation by Using a Temporary Crown as a Master Template
The Epimine Resin Temporary Crown
Construction of an Epimine Resin Temporary Crown
Finishing the Typical Maxillary Central Incisor Preparation
Micro-diamond finishing
Tungsten-Carbide Finishing “Stones”
Polishing the Preparation
Pre-packing of the gingiva
Common Causes of Fracture in Porcelain Jacket Crowns
The Metal-Ceramic Veneer Crown
Reduction of stress and creep in the metal porcelain combination
The Labial Shoulder
Optimising the aesthetics and maintaining the marginal fit on the labial and buccal porcelain
The shoulder versus the bevel
Effect of burnishing
The gold collar and bevel
The Shoulder and Chamfer Preparation
Influence of shoulder design on Metal Creep
The Approximal and Lingual Shoulders
Recommended Designs for the Preparation and Metal Coping in Metal-Ceramic Crowns
Labial Shoulder
Chamfer with bevel
Lingual Shoulder
Recommended designs for the preparations and metal coping
The Metal-Ceramic Crown
Typical Maxillary Central incisor Preparation. Operative Procedures
Typical Maxillary Premolar Preparation for Full Porcelain Veneer Coverage
Preparation of the Typical Maxillary Premolar for Partial Porcelain Veneer Coverage
Improving Retention of Metal-Ceramic Crowns
Advantages of Partial Veneer Coverage of Porcelain over Full Veneer Coverage
Disadvantages of Partial Veneer Coverage of Porcelain over Full Veneer Coverage
Preparation for the periodontally involved tooth
Common Faults in Preparation
Biological Considerations
The Impression
Relationship of the gingival tissue to the crown or margin
Selection of Impression Materials and Techniques
The Copper Tube Impression
The Transfer Coping
The Elastic Impression
Surface Reproduction
Compatibility with Model Materials
Dimensional Stability
Coefficient of Thermal Expansion
Elasticity
Tearing Energy
Cost
Types of Elastic Impression Material
Reversible Hydrocolloid
Polysulphide Rubber
Silicone Rubber (Organo tin catalysts)
Silicone Rubber (Chloroplatinicacid catalyst)
Polyether Rubber
Evaluation of Elastic impression Materials
Clinical Recommendations
Procedures for taking Polysulphide or Polyether Elastomeric Impressions
Requirements for a Custom-Built Tray
Preparation of the Gingival Tissue
Mechanical Retraction
Mechanical-chemical Retraction
Technical considerations
Gingival packing instrument
Preparation of Shoulder areas
Surgical Retraction
Steel scalpel and electrosurgical cutting on gingival tissues and alveolar bone
Factors governing optimal electrosurgical cutting
Impression Technique
Choice of syringe
Alternative methods
Common Causes of Failure in Elastomeric Impression Materials
Surface Defects
Dimensional Instability
Cementation of Porcelain and Metal-Ceramic Crowns
Recommended clinical use for dental cements
Cementation of crowns with good retentive form and where pulpal damage may occur
Cementation of crowns with moderate retentive form and where pulpal damage may occur
Cementation of crowns where no possible damage to the pulp can occur
Cementation of complete porcelain crowns
Cementation of post crowns, facings or crowns on metal substructures
Cementation of fixed splints or bridgework
Clinical Manipulation
References
Appendix
Introduction
Ceramics are the earliest group of inorganic materials to be structurally modified by man, and his early history is principally traced through these materials. The origin of glazing techniques is probably the most interesting advance that was later to be so significant from a dental standpoint. Glazed porcelain is our only restorative material from which bacterial plaque can be easily removed, and increasing attention is now being paid to research into the significance of plaque formation on dental restorations.
The earliest glazing technique was a Sumerian invention made famous about 4000 B.C. as Egyptian blue faience. This glaze was not, like later ones, a melted premix of glass-forming materials but was made by a type of cementation process. Potash was drawn by capillarity to react with the surface of a preformed body of siliceous particles to form a glassy coat of copper-coloured eutectic silicate. The process is still in use today in Iran.
From this early work stemmed all the developments in ceramic technology, and the remarkable fact is that the early ceramists were exploiting almost all the properties of solids that are the concern of the modern solid-state physicists. With the exception of electrical and magnetic effects the ceramist was, sometimes inadvertently, using such properties as moisture-dependent plasticity and thixotrophy in his shaping techniques. Decorative textures were derived from vitrification and devitrification, the nucleation of various crystalline
phases, and local variations of viscosity, surface tension and expansivity. Colours depended on various states of oxidation, on abnormal ionic states, on excitons and on structural imperfections in crystals. The understanding of these processes came later when crystal structures could be analysed by means of X-ray diffraction. Their complexity is such that even today much of our decorative ceramic technique remains an art rather than a science. Equally the building of a porcelain jacket crown requires artistic skill; however, with an understanding of the science of ceramics, the technician is better placed to improve his art.
The European development of porcelain came about in the 18th century, and the originator of the first porcelain paste used for denture work was a French apothecary, Alexis Duchâteau. His early dentures were ill-fitting because of the uncontrolled firing shrinkage, and after several fruitless experiments, he sought the help of a dentist, Dubois de Chemant, who had all the qualities of the tireless clinician. In 1788, de Chemant published his book on artificial teeth and the subsequent action brought against him by Parisian dentists who accused him of stealing Duchâteau’s invention led to his emigration to England.
John Woodforde in his book “The Strange Story of False Teeth” describes how de Chemant set up in London in 1792 and continued to manufacture dentures of porcelain paste supplied by the famous Wedgwood porcelain factory.
The first single porcelain teeth were launched in 1808 by an Italian dentist, Guiseppangelo Fonzi, who worked in Paris, but they never met with great approval because of their brittleness and opacity. It was not until the 1850’s that Samuel Stockton of Philadelphia, his nephew S. S. White, and Claudius Ash, in England, placed the porcelain tooth on a successful commercial basis. Then with the advent of vulcanite rubber at this time, dentures for the masses became a reality.
The dental profession did not really master the art of ceramics until the end of the nineteenth century when fixed restorations were realised. Dr. Charles H. Land of Detroit was a pioneer in this field and he filed the first patent in 1889 for the construction of the porcelain jacket crown. An ever-growing list of clinicians followed in his footsteps, and the refinement of the shoulder preparation in 1903 has beeh attributed to Dr. E. B. Spaulding. Dr. W. A. Capon of Philadelphia and Dr. Hugh Avery of San Francisco, did much to enhance the porcelain inlay technique and indeed some of the concepts advanced at that time still find a place in text-books today.
The old trick of baking a porcelain jacket crown to fit a nail was reported in 1908 and Dr. A. E. Schneider was impressing audiences as he hammered the crown into a block of wood. Schneider, at that time, also made the observation that the crown shoulder should be at a right angle to the force of occlusion. With the publication of Dr. Albert Le Gro’s book on “Ceramics in Dentistry” in 1925, the use of porcelain had become firmly established. About this time in Europe, the high fusing porcelains were developed to a high degree of aesthetics and Jan Adriaansen of Amsterdam pioneered the technique of building up porcelain with a brush. He also developed “Prisma” high fusing porcelain with Harrison’s, the glaze manufacturers at Stoke-upon-Trent in England, many of these crowns being in use today.
During this period, several attempts were made to use metal reinforcement in the porcelain. Iridioplatinum was used in various forms by Doctor Swann, Felcher, Hovestad, Johnson, Lakermance, Gonod and Granger, and their pioneering of the reinforced porcelain bridge led to the development of the current metal-ceramic materials.
It was not until 1962, when M. Weinstein, S. Katz and A. B. Weinstein filed their first patent in the U.S.A. on the use of gold alloys for porcelain bonding that the universal use of metal-ceramics became a real possibility. Other methods of reinforcing porcelain were also being explored at this time, and the first viable technique for making alumina reinforced crowns was developed in 1963 by McLean and Hughes in England. This work has been taken a stage further by McLean and Sced in 1976 in the development of a stronger platinum bonded alumina crown. Attachment of aluminous porcelain to the platinum being achieved by surface coating of the metal with a thin layer of tin. This new system of porcelain attachment using electroplating techniques does not require base metals to be incorporated in the alloy as in the previous metal-ceramic systems.
Other major advances were made in firing dental ceramics and Vines, Semmelman, Lee and Fonvielle developed the use of vacuum firing techniques at the Dentists’ Supply Company in the U.S.A. This method opened up a whole new field in aesthetic dentistry.
With the introduction of all these new porcelains and techniques, the current interest in dental ceramics is growing even faster. Dental porcelain has now reached a stage of development where it seems highly unlikely to be replaced by plastics within the foreseeable future. Materials science is expanding quickly and new methods of strengthening glasses, including techniques such as nucleation, ion-exchange and dispersion strengthening of glasses have yet to be fully exploited in dentistry.
It is very probable that the dental profession will be using glasses or ceramics well into the next century for the replacement of lost tooth enamel. Even assuming that the problem of caries is conquered, we must be ever mindful of the effects of natural abrasion on tooth structure and it is likely that this problem could become of major concern to a longer-living populace demanding retention of its teeth.
References
Avery, H. (1916). Present Status of Porcelain in Dentistry. Paper to the Annual Session N.D.A. July.
Felcher, F. R. (1937). Platinum Reinforced Porcelain Restorations, D. Digest 43:24.
Granger, E. R. (1940). Platinum-iridium Castings; a new Concept of the Dental Casting Process. J.A.D.A. 27:1718.
Hovestad, J. F. (1936). Porcelain Bridges. D. Items Int. 58:239, 330, 418, 509, 615.
Lakermance, R., and Gonod, P. Bridges in Reinforced Porcelain. J. Am. D. Club Paris 2:1.
Le Gro, A. L. (1921). Present Status of Porcelain in Dentistry. Paper to the Michigan State Dental Society. April.
Le Gro, A. L. (1925). Ceramics in Dentistry, Dental Items of Interest Publishing Co. Brooklyn, N. Y.
McLean, J. W., and Hughes, T. H. (1965). The Reinforcement of Dental Porcelain with Ceramic Oxides. Brit. dent. J. 119:251.
McLean, J. W., and Sced, I. R. (1976). The Bonded Alumina Crown I. The bonding of platinum to aluminous dental porcelain using tin-oxide coatings. Austr. Dent. J. 21, 119.
McLean, J. W., and Sced, I, R. (1977). Production of Metal-Ceramic Dental Restorations. British Pat. No. 1,483,362.
McLean, J. W., and Hughes, T. H. (1968). Improvements in Dental Materials. British Pat. No. 1,105,111.
Schneider, A. E. (1908). Paper Presented to the Illinois State Dental Society.
Smith, C. S. (1967). Materials. Scient. Amer. Sept. 217:69.
Swann, H. (1931). Torque Resisting Porcelain Bridge, Chicago.
Tylman, S. D. (1970). Theory and Practice of Crown and Fixed Partial Prosthodontics (bridge). C. V. Mosby Co., St. Louis.
Vines, R. F., Semmelman, J. O., Lee, P. W., and Fonvielle, F. D. (1958). Mechanisms Involved in Securing Dense Vitrified Ceramics from Preshaped Partly Crystalline Bodies. J. Amer. Ceram. Soc., 41:304.
Weinstein, M., Katz, S., and Weinstein, A. B. (1962). U.S. Pat. No. 3,052,982.
Monograph I
The Nature of Dental Ceramics
The word “ceramic” is derived from the Greek “keramikos” which means “earthen”. A ceramic is therefore an earthy material, usually of a silicate nature and may be defined as a combination of one or more metals with a non-metallic element, usually oxygen (Gilman, 1967). The larger oxygen atoms serve as a matrix, with the smaller metal atoms (or semi-metal atoms such as silicon) tucked into the spaces between the oxygens (Fig. 1-1).
The atomic bonds in a ceramic crystal have both a covalent and ionic character. These strong bonds are responsible for the great stability of ceramics and impart very useful properties, such as hardness, high modulus of elasticity and resistance to heat and chemical attack. On the other hand, the nature of this bonding creates difficulties for the dental ceramist since all ceramic materials are brittle.
Crystalline Ceramics
Regular dental porcelain, being of a glassy nature, is largely non-crystalline, and exhibits only a short range order in atomic arrangement. It is therefore more appropriate to consider this material in the second part of this chapter, “The Nature of Glasses”.
The only true crystalline ceramic used at present in restorative dentistry is Alumina (Al2O3) which is the hardest and probably the strongest oxide known. The hardness and strength of alumina makes it difficult to cleave because of the “interlocking” nature of the structure. Binns (1970) considers that when a plane intersects a crystal, the force holding the two halves of the crystal together depends upon the number of chemical bonds cut and the strength of the individual bonds. The number of bonds is dependent upon the packing of the solid and is given by the number of gram-atoms per unit volume. The bond strength is less easy to assess, but probably the most important feature is the extent to which bonding is covalent. Ionic potential is defined as charge/ionic radius and Pauling (1945) has reported values. The greater the ionic potential of a cation the greater its polarizing power on the anion. According to Fajans (Weyl and Marboe, 1962) the more the anion is polarized by the cation the greater is the degree of covalency of the bond. Thus the degree of covalency, and hence the bond strength, increases with the valency. In the series of oxides Na2O, MgO, Al2O3, SiO2, where the ionic potential of the cation increases, the bonding becomes increasingly covalent. In the case of Na2O the bonding is almost completely ionic, whereas SiO2 would have about 50 percent covalent bonding. If the number of gram-atoms is multiplied by the valency of the metal, a quantity is obtained which, for some of the harder materials, compares reasonably well with the hardness. This is a simplified explanation which applies only to perfect single crystals; in practice, the conclusions are modified by dislocations, slip, grain boundary effects, and flaws.
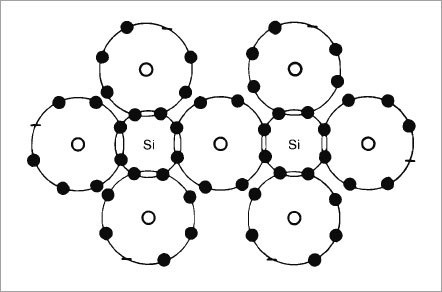
Fig. 1-1a Diagram of a silicate unit with each SiO tetrahedra sharing an oxygen atom.
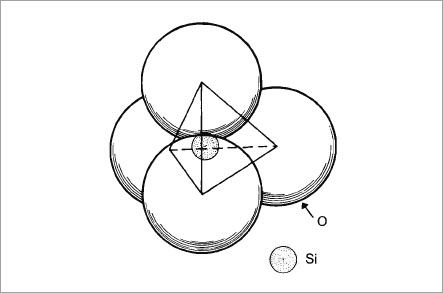
Fig. 1-1b Three dimensional drawing of a silicate unit in which the silicon atom Si is surrounded by four oxygen atoms.

Fig. 1-1c Three dimensional drawing of linked silicate units which form the continuous network in glass.
Extraction of Alumina
Alumina is the oxide of aluminium (Al2O3), commonly extracted from the mineral bauxite, which is mainly a hydrated aluminium oxide. According to normal practice, the ore is crushed and ground to – 10 mesh and is digested in a concentrated solution of caustic soda. The aluminium-bearing liquor recovered from this process is clarified, and the alumina is precipitated in the form of alumina trihydrate crystals which are then washed and dried without removal of the chemically combined water. The alumina trihydrate is converted to alumina by calcination, usually in a rotary kiln at a temperature of 600°C which drives off the chemically combined water in the hydrate to form gamma-alumina. Further calcination at 1250°C converts it to alpha-alumina (gamma-alumina is predominently required by American metal producers while European users mainly require the alpha-form). For ceramic applications the alpha-form is employed and is generally ball milled and commercially supplied as a fine powder, usually below 10 to 20 microns in size.

Fig. 1-2 Sintered high alumina profiles used in restorative dentistry.
Fabrication of Alumina Components
Sintered alumina is used in restorative dentistry in the form of prefabricated profiles or reinforcements for the construction of crowns, bridges, or individual pontics (McLean and Hughes, 1965).
These reinforcements are made by mixing the fine calcined alumina powder with a binder such as methyl cellulose and a release agent. The plastic alumina mass is then extruded through tungsten carbide nozzles to the desired shape or form; for dental purposes rods, tubes or sheets are most commonly used (Fig. 1-2).
The moulded profiles are then placed on refractory trays and fired in a standard industrial tunnel kiln. Very slow oven drying is used to prevent warping and the alumina is finally sintered or recrystallised at temperatures of up to 1650°C. The resulting product is a hard, impermeable ceramic of very high strength and chemical resistance.
Sintering of Alumina
Many theories exist as to the exact processes which occur during the firing cycle of alumina, although none are entirely certain; the terms sintered, fused and recrystallised are widely used to describe the alumina end-product.

Fig. 1-3 Photomicrograph showing the surface of sintered alumina (95 percent Al2O3). Relief polished. Normaski interference contrast. Mag x 850. Courtesy of D. B. Binns. British Ceramic Research Association.
Burke (1958) defined the term “recrystallisation” as changes in microstructure that occur in crystalline or largely crystalline bodies when atoms move to positions of greater stability. By contrast metallurgists use the term recrystallisation in a much more restricted sense: the nucleation and subsequent growth of a new generation of strain-free grains into the deformed matrix of a cold-worked material.
It appears that during the firing of alumina, the following steps occur. Firstly, a welding occurs at points of contact between adjacent oxide particles, giving rise to a lensing effect as normally occurs in sintering processes, i.e. partial fusion. Migration of atoms then leads to growth of the lens areas, movement of grain boundaries and reduction in porosity. During sintering, the shift in grain boundaries results in the formation of a closely interlocking crystalline structure of considerable strength (Fig. 1-3). This improved packing of the oxide particles results in shrinkage of the ceramic body and compensatory mould design is required similar to the oversize tooth moulds used in a tooth factory.
The driving force for the shrinkage in alumina ceramics is surface tension. The surface tension of free surfaces (pores etc.) in a porous ceramic body will always try to make the piece shrink to reduce surface energy. It has been suggested by Nabarro (1948) and Herring (1950) that the lattice vacancies formed at the surface of a pore can be discharged at grain boundaries as well as at the free surface of the piece, and this might explain the relative independence of sintering rates upon specimen size. Sintered alumina will exhibit this phenomenon and it can be shown that pores near the grain boundaries have disappeared, whereas pores near the centre of the grains remain (Fig. 1-4). The latter pores account for the opacity of alumina where only 2 to 10 percent light transmission is obtainable on 1 mm thick discs (McLean, 1966). This opacity will therefore make alumina suitable for use only in the anchorage areas of artificial teeth or crowns.
Translucent Alumina
More recently methods of firing high purity aluminas containing up to 0.2 percent MgO have been devised in which a spinel is formed at the grain boundaries, slowing down grain growth and allowing the diffusion of porosity along grain boundaries. These aluminas (G.E.C. Lucalox) are probably fired in hydrogen or oxygen atmosphere at high temperature (1800°C) which increases rate of diffusion of porosity. The resultant materials are almost pore-free which in turn produces a highly translucent body, through which it is possible to read newsprint. It is conceivable that these materials might have possibilities in a veneer crown technique where the alumina is used as the main reinforcing shell.
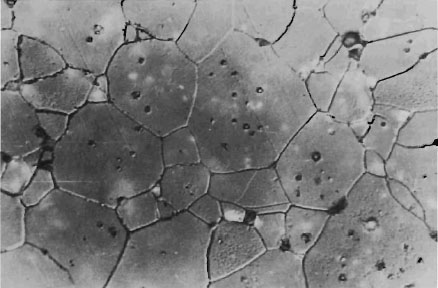
Fig. 1-4 Photomicrograph of pores trapped in the centre of sintered alumina crystals (99.5 percent Al2O3). Thermal etch. Normaski interference contrast. Mag × 775. Courtesy of D. B. Binns. British Ceramic Research Association.
Effect of Debasing Alumina
The function of a debasing or fluxing agent, added to high purity alumina, is to lower the sintering temperature. It may do this either by forming a liquid phase, or by going into solid solution in the alumina lattice and increasing the diffusion rate.
Debasing agents must be selected so that they do not appreciably affect the mechanical properties of the fired specimens; for example, by causing excessive grain growth which weakens the ceramic body.
A wide range of compositions has been used in the past, but the chief oxides used are CaO, MgO, SiO2, TiO2, MnO2. The function of the first three is almost entirely that of forming a glass phase to bond the structure together. The last two, although also taking part in glass formation, are generally added to increase atomic diffusion in the alumina itself. The proportions of the different oxides used has varied over quite a wide range, but it is probably most common for the SiO2 content to be at least half of the added oxide content and for the CaO content to be larger than that of MgO. Useful ways of adding these oxides are as wollastonite (Ca Si O3) and talc (Mg3Si4O10(OH)2).
A high purity alumina of 97 to 99 percent requires a firing temperature of 1600°C to 1650°C, for a mixture which has been debased to 85 percent purity of alumina, the balance being at least half SiO2; sintering occurs at 1500°C, whilst a mixture comprising 75 percent alumina may be fired at 1300°C to 1350°C. This enables cheaper and simpler furnaces to be used for the firing process. A typical composition for an 85 percent alumina refractory mixture would be as follows:
| Weight percent | ||
| Alumina | Al2O3 | 87.70 |
| Silica | SiO2 | 7.10 |
| Calcium Oxide | CaO | 1.61 |
| Magnesium Oxide | MgO | 1.32 |
| Chromium Sesquioxide | Cr2O3 | 1.13 |
| Ferric Oxide | Fe2O3 | 0.25 |
| Titanium Dioxide | TiO2 | 0.13 |
| Sodium Oxide | Na2O | 0.40 |
| Potassium Oxide | K2O | 0.36 |
Colouring Alumina
The colour of sintered alumina is white or cream. However, in order to use this material in restorative dentistry, a range of suitable background colours is necessary when enamel veneers are applied. Dental alumina is therefore coloured with high temperature resistant pigments. These pigments may consist of up to 2 percent of manganese-alumina pink, vanadium-zircon blue, or praesodymium-zircon yellow (McLean, 1966).
Dental Alumina
Composition. Alumina ceramics used in dentistry are of high purity and generally consist of at least 95 percent Al2O3. Minor quantities of debasing agents are added to aid sintering, and pigments of the type just mentioned are added to produce a natural dentine colour. Sintered alumina ceramics of high purity are conveniently referred to as high alumina.
Physical Properties
The physical properties and characteristics of a dental high alumina are illustrated in Table 1-1. These high purity aluminas have excellent resistance to abrasion and chemical attack and are suitable for use up to a working temperature of 1500°C. The significance of this is that at normal firing temperatures used in dental furnaces (900°C to 1150°C) the alumina is completely stable and will not creep or flow. For purposes of comparison the mechanical properties of dental porcelain have been included in Table 1-1 and it may be seen that high alumina is markedly stronger in all essential properties. The high tensile strength is perhaps of greatest significance since tensile failure of dental ceramics is the commonest cause of fracture. Although the strength of high alumina is quite remarkable for a ceramic material, it still does not match its theoretical strength.
The prime causes of weakening in crystalline ceramics has been well summarized by Gilman (1967).
It is clear that all high strength ceramics suffer more from crystal boundary defects than metals, and current research is concentrating on eliminating internal boundaries or minimising their effects.
The use of ceramics in fixed bridgework is limited by these inherent physical defects and in addition shrinkage control still remains one of the main problems with crystalline ceramics. The versatility of metals both in casting accuracy and the nature of the metallic bond leaves them unrivalled in restorative dentistry.
| High Alumina | Dental Porcelain | ||
| Tensile Strength | 138N/mm2 | (20,000 p.s.i.) | 34.4 N/mm2 (5,000 p.s.i.) |
| Compressive Strength | 2,180 N/mm2 | (316,000 p.s.i.) | 344 N/mm2 (50,000 p.s.i.) |
| Modulus of Rupture | 379 N/mm2 | (55,000 p.s.i.) | 68.9 N/mm2 (10,000 p.s.i.) |
| Hardness Moh Scale | 9 | ||
| Coefficient of Linear Expansion Parts/million/°C | |||
| 20°C – 400°C | 6.6 | ||
| 20°C – 600°C | 7.2 | ||
| 20°C – 1,000°C | 8.2 | ||
| Porosity Fuchsine dye penetration | Nil | ||
| Water absorption | Nil | ||
| Specific gravity | 3.85 | ||
| Working temperature maximum °C | 1,500 | ||
| Colour | Ivory | ||
The Nature of Glasses
Dental porcelains are, in part, glassy materials and in order to understand their formulation, a knowledge of glass formation is essential. Glasses may be regarded as supercooled liquids or as non-crystalline solids. This lack of crystallinity distinguishes them from other solids, and their atomic structures and resultant properties depend, not only on composition, but also on thermal history. The glass-maker selects complex or impure solutions in order to modify the physical properties of the glass both in the molten and solid form. These properties would include viscosity, melting temperature, chemical durability, thermal expansion and resistance to devitrification. Frictional forces inhibiting the formation of new molecular configurations in the liquid glass should also be as high as possible. This can be achieved by rapid cooling or preferably, for dental purposes, by using materials that produce high viscosity in the melt, e.g. Aluminium oxide Al2O3.
Glass Formation
The principal anion present in all glasses is O2− ion which forms very stable bonds with small multivalent cations such as silicon, boron, germanium, or phosphorous, giving rise to structural units, such as the SiO4 tetrahedra illustrated in Fig. 1-1, which form a random network in glass. These ions are thus termed glass formers.
Zachariasen (1932) examined the characteristics of these glass-forming oxides and proposed his random network theory of glass structure. He considered that the interatomic forces in glasses and crystals must be essentially similar and that the atoms in glass oscillate about definite equilibrium positions. He deduced that the atoms must be linked in the form of a three-dimensional network in glass as in crystals, and because glasses do not give a sharp X-ray diffraction spectra they could not be periodic (in an ordered arrangement). Zachariasen considered that although the units of structure in the glass, (i.e. SiO4 tetrahedra) and in the crystals are practically identical, in the crystal these structural units are built up to give a regular lattice. By contrast, in the glass there is sufficient distortion of bond angles to permit the structural units to be arranged in a random network (Fig. 1-5). Zachariasen proposed certain conditions for glass formation:
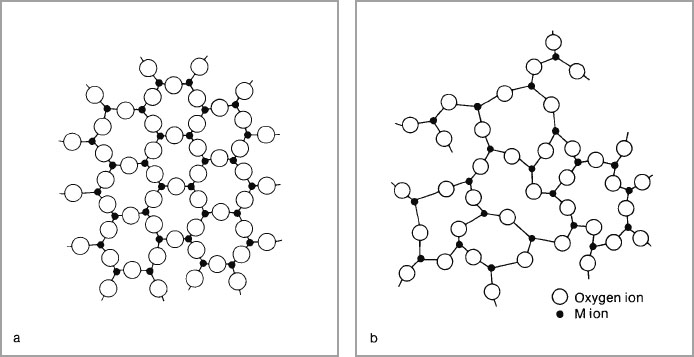
Fig. 1-5a and b Two-dimensional representation of an oxide M2O3 in (a) the crystalline form (b) the glass form. Adapted from McMillan, P. W. Glass-Ceramics (1964) Academic Press.
For dental purposes, only two glass-forming oxides are used – silicon and boron oxide – and they form the principal network around which a dental glass can be built. Alumina, under certain circumstances, may be regarded as a glass-forming oxide when in combination with other oxides as will be explained.
Formulation of Dental Porcelain
Dental porcelains use the basic silicon-oxygen network as the glass-forming matrix but additional properties, such as low-fusing temperature, high viscosity, and resistance to devitrification, are built in by the addition of other oxides to the glass-forming lattice SiO4. These oxides generally consist of Potassium, Sodium, Calcium, Aluminium and Boric oxides.
The Role of Fluxes
Potassium, Sodium and Calcium oxide are used as glass modifiers, and act as fluxes by interrupting the integrity of the SiO4 network. The purpose of a flux is principally to lower the softening temperature of a glass by reducing the amount of cross-linking between the oxygen and glass-forming elements, e.g. silicon. For example, if soda (Na2O) is introduced into a silicate melt to produce sodium silicate glasses, structural changes occur as in Fig. 1-6. Instead of the bridging oxygen ions which formed the link between the two SiO44Fig. 1-7+